Department of Precision Health
At the DoPH, we implement interdisciplinary approaches at the convergence of precision medicine, prevention and public health. We combine established and new methods with novel digital technologies for epidemiological and public health research. Through translating our results into clinical practice and supporting public health bodies, we aim at tackling the major causes of morbidity and mortality, improving quality of life and delivering scientific evidence for personalized disease prevention and intervention.
Vision & Mission
Our expertise spans a wide range of disciplines including epidemiology, public & digital health, health economics, (neuro)psychology, molecular biology, biochemistry and toxicology. We study the exposome (environmental factors, nutrition, physical activity) and its determinants (e.g sociodemographic inequalities), but also omics (e.g miRNA) and digital biomarkers (ePROs, vocal and movement-based fingerprints, data from medical and connected devices).
We use both hypothesis- and data-driven approaches to analyse large multi-dimensional datasets (observational, epidemiological and clinical cohort studies, real-world data, health registries, electronic health records,…), leveraging both conventional and innovative approaches (e.g. digital devices, web-based sources, Artificial Intelligence (AI)). This broad expertise enables us to obtain a 360°-view of an individual. This notably allows a finer stratification of patients and populations based on various biological, clinical, environmental and lifestyle information.
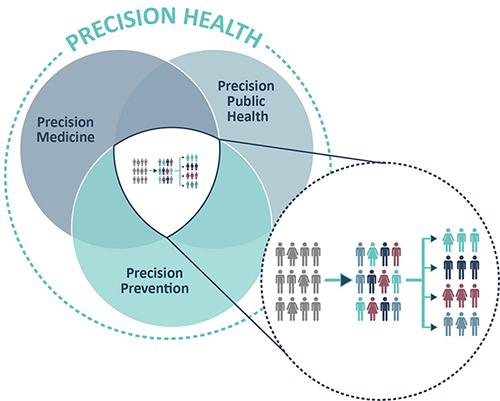
The goal of our interdisciplinary team is to foster the integration of digital technologies and real-world data into public health, translational research and clinical practice for the benefit of all. To achieve this, we develop collaborative research projects with national and international research institutions, clinicians, companies, patient associations and policymakers. We work on various conditions including cancer, cardiometabolic and neurodegenerative diseases. We also support public health bodies in setting up and maintaining disease registries, conducting national surveys and developing policies.
We ultimately aim to translate our findings to personalize prevention, treatment and care in order to deliver the right intervention for the right individual or population at the right time.
Director of Department

FAGHERAZZI
- Ageing, Cancer, and Disparities Research Unit – ACADI
- Brain Imaging & Neuro Epidemiology, DoPH
- Cardiovascular Research Unit
- Deep Digital Phenotyping Research Unit
- Human Biomonitoring Research Unit
- Human Motion, Orthopaedics, Sports Medicine and Digital Methods
- NutriHealth
- Physical Activity, Sport and Health
- Public Health Expertise
- Socio-Economic & Environmental Health & Health Services
WATCH 📺 – PRECISION HEALTH RESEARCH AT DOPH
The DoPH focuses its interdisciplinary research around the vision of Precision Health, encompassing aspects of precision public health, innovative exposure measurements (hair analysis, new biomarkers, etc.), precision prevention, lifestyle, health economics and digital health. The ultimate objective of the department is to provide more tailored, personalized prevention and treatment. In the current digital and data-driven world, to achieve this the researchers can rely on an increasing volume of data generated by populations and patients. The DoPH is strongly engaged in patient-centric research using new technologies and big data.
Watch the thematic videos to learn more about DoPH research activities in the domain of precision health. The talks were recorded during a DoPH webinar series dedicated to Precision Public Health in 2021.
Projects & clinical trials
Featured team members
Scientific publications
-
Shared splicing dysregulation in heart failure associated with dilated and ischemic cardiomyopathy and spatial specificity across cardiac regions – 24/03/2026
Sandra Martins and 10 other authors -
Progression of
GBA1 severe and risk variants – 26/03/2026
Anne-Marie Hanff and 12 other authors -
Empowering healthy lifestyle behaviour through personalised intervention portfolios using a healthy lifestyle recommender system to prevent and control obesity in older adults – 25/03/2026
Torsten Bohn and 22 other authors -
Ten-Year Trends in Healthcare Use and Costs for People Living with Diabetes in France – 25/03/2026
Guy Fagherazzi and 9 other authors -
Association of eating disorders and/or insulin omission with impaired glycaemic control in persons living with type 1 diabetes – 19/03/2026
Patrick Jean Ritz and 24 other authors -
Translating AI research into reality – 16/03/2026
Samantha Salvi Cruz and 49 other authors -
Pros and cons of Swedish law on intellectual property rights – 13/03/2026
Ljubica Matic and 2 other authors -
Voice Signatures of Momentary Psychological Stress in Real-Life Environments – 01/01/2026
Guy Fagherazzi and 6 other authors -
Time to modernise research in geriatric oncology – 13/12/2025
Sophie Pilleron -
Interactions between genetic predisposition to obesity, insulin resistance and type 2 diabetes risk, and food or beverage intake for incident type 2 diabetes – 01/03/2026
Nicholas J. Wareham and 31 other authors -
Ethical Challenges in Automated Insulin Delivery and Emerging Diabetes Technologies – 01/02/2026
Ralph El Khoury and 2 other authors -
Multimodal (Bio)Markers and Risk of Obesity – A Comprehensive Scoping Review – 24/12/2025
Torsten Bohn and 19 other authors -
Psychosocial burden in type 1 diabetes – 29/01/2026
Gloria Aguayo and 10 other authors -
Do nurses and allied professionals need to upskill to meet developments in RNA therapeutics for CVD? – 09/01/2026
Faye Forsyth and 1 other author -
Direct RNA sequencing identified solute carrier family 2 member 1 to improve neurological outcome prediction after cardiac arrest – 07/01/2026
Yvan Devaux and 16 other authors -
Defining and addressing loneliness in older adults with cancer – 01/01/2026
Enrique Soto-Perez-de-Celis and 12 other authors -
Empowering Healthy Lifestyle Behavior Through Personalized Intervention Portfolios Using a Healthy Lifestyle Recommender System to Prevent and Control Obesity in Young Adults – 13/12/2025
Silvia Sánchez-García and 16 other authors -
An app-based physical activity intervention for people with hip and knee osteoarthritis – 05/12/2025
Laurent Malisoux and 4 other authors -
Association between transportation noise exposure and type 2 diabetes risk in a French prospective cohort – 02/12/2025
Elodie Faure and 16 other authors -
Severity of complications is associated with impaired health-related quality of life in people with type 1 diabetes – 01/01/2025
Sara Barraud and 20 other authors -
Beyond overweight, visceral adiposity is associated with estimation of cardiovascular risk in patients living with type 1 diabetes – 01/12/2025
Laurence Salle and 17 other authors -
Time below range alone is insufficient to identify severe hypoglycaemia risk in type 1 diabetes—the critical role of hypoglycaemia awareness – 09/09/2025
Dulce Canha and 15 other authors -
A nationwide 12-month observatory of automated insulin delivery shows improved glucose control, sustained adoption, and reduced acute severe events – 01/01/2025
J. P. Riveline and 49 other authors -
Chemical pollutant mixtures associated with metabolic health – 10/10/2025
Jesús Martínez Gómez and 6 other authors -
Co-occurrence of memory impairment and fatigue distinguishes post COVID from pandemic-related health effects in the 4-year CON-VINCE cohort study – 27/10/2025
Dmitry Bulaev and 85 other authors -
Bottlenecks in advancing and applying multiomic data integration-common data resources as rate-limiting drivers-the high-impact use case of atherosclerotic cardiovascular disease – 10/10/2025
Stephanie Bezzina Wettinger and 22 other authors -
Nationwide Trends in Type 1 and Type 2 Diabetes in France (2010–2019) – 01/01/2025
M. Joubert and 9 other authors -
Geographic environments, daily activities and stress in Luxembourg (the FragMent study) – 02/09/2025
Camille Perchoux and 14 other authors -
Established and Emerging Roles of Epigenetic Regulation in Diabetic Cardiomyopathy – 01/09/2025
Adam Russell-Hallinan and 11 other authors -
Multiomics in atherosclerotic cardiovascular disease – 26/06/2025
Liv Tybjærg Nordestgaard and 12 other authors -
How much running is too much? – 01/01/2025
Jesper Schuster Brandt Frandsen and 12 other authors -
Role of the exposome in mental disorders – 18/08/2025
Claudia Gutierrez Ortiz and 4 other authors -
Immune cell–adipose tissue crosstalk in metabolic diseases with a focus on type 1 diabetes – 04/06/2025
Rasheed Ahmad and 6 other authors -
Voice Assessment and Vocal Biomarkers in Heart Failure – 24/04/2025
Fabian Kerwagen and 10 other authors -
A Functionally-Grounded Benchmark Framework for XAI Methods – 14/07/2025
Dulce Canha and 3 other authors -
Designing and Developing a Population/Literature-Based Westernized Diet Index (WDI) and Its Relevance for Cardiometabolic Health – 14/07/2025
Miguel Cifuentes and 3 other authors -
Vitamin A5 – 14/07/2025
Ralph Rühl and 8 other authors -
Prognostic and predictive microRNA panels for heart failure patients with reduced or preserved ejection fraction – 07/07/2025
Reza Parvan and 8 other authors -
Musculoskeletal disorders in type 1 diabetes – 23/04/2025
Noémie Topalian and 19 other authors -
Racial disparities in continuous glucose monitoring-based 60-min glucose predictions among people with type 1 diabetes – 30/06/2025
Adam Hulman and 8 other authors -
Daily mobility, activity and environmental determinants of stress in ecological momentary assessment (EMA) and GPS studies – 27/06/2025
Noémie Topalian and 2 other authors -
Master protocols in vocal biomarker development to reduce variability and advance clinical precision – 27/06/2025
Yaël Bensoussan and 4 other authors -
Editorial – 01/01/2025
Miron Sopic and 4 other authors -
Protocol for an umbrella review of systematic reviews evaluating the efficacy of digital health solutions in supporting adult cancer survivorship care – 27/05/2025
Danielle Keane and 60 other authors -
Strengthening interprofessional and interdisciplinary collaboration in geriatric oncology initiatives – 30/04/2025
Fay J. Strohschein and 10 other authors -
Trajectories of persisting Covid- 19 symptoms up to 24 months after acute infection – 25/04/2025
Aurélie Fischer and 8 other authors -
Cross-sectional analysis of eating disorder risk and risk correlates in candidates for bariatric surgery from the BariPredict cohort – 01/04/2025
Fawaz Alzaid and 11 other authors -
The effect of automated insulin delivery system use on diabetes distress in people with type 1 diabetes and their caregivers – 26/12/2024
Gloria Aguayo and 7 other authors -
The role of healthcare professionals’ attitudes in treatment decision-making for older adults with cancer – 01/01/2024
India Pinker and 3 other authors -
Interactive Panel Summaries of the 2024 Voice AI Symposium – 27/03/2025
Jean Christophe Bélisle-Pipon and 32 other authors -
Association of LEF1-AS1 with cardiovascular and neurological complications of COVID-19 – 22/12/2024
Yvan Devaux and 8 other authors -
Epidemiology of cancer in older adults – 01/01/2025
Esther Bastiaannet and 1 other author -
Moving beyond studying mere association – 01/01/2024
Sophie Pilleron -
Clinical phenotyping of people living with type 1 diabetes according to their levels of diabetes-related distress – 12/08/2024
Guy Fagherazzi and 15 other authors -
Models of care and associated targeted implementation strategies for cancer survivorship support in Europe – 16/02/2025
Gillian Prue and 38 other authors -
Age-related differences in staging, treatment and net survival in relation to frailty in adults with colon cancer in England – 02/02/2025
Eva J.A. Morris and 4 other authors -
Blood CD45
+/CD3
+ lymphocyte-released extracellular vesicles and mortality in hospitalized patients with coronavirus disease 2019. – 01/01/2024
Lina Badimon and 12 other authors -
Readiness of adults with type 1 diabetes and diabetes caregivers for diabetes distress monitoring using a voice-based digital health solution – 02/01/2025
Gloria Aguayo and 8 other authors -
EDITORIAL for BJP themed issue “noncoding RNA therapeutics” – 21/11/2024
Yvan Devaux and 2 other authors -
A voice-based algorithm can predict type 2 diabetes status in USA adults – 19/12/2024
Guy Fagherazzi and 8 other authors -
Estimated Dietary Intakes of Vitamin A5 – 22/11/2024
Ralph Rühl and 3 other authors -
The Imperative of Voice Data Collection in Clinical Trials – 13/11/2024
Guy Fagherazzi and 1 other author -
Worldwide trends in diabetes prevalence and treatment from 1990 to 2022 – 13/11/2024
Bin Zhou and 49 other authors -
Running shoe cushioning properties at the rearfoot and forefoot and their relationship to injury – 11/10/2024
Laurent Malisoux and 4 other authors -
Digital voice-based biomarker for monitoring respiratory quality of life – 01/10/2024
Vladimir Despotovic and 6 other authors -
Dopamine Pathway and Parkinson’s Risk Variants Are Associated with Levodopa-Induced Dyskinesia – 01/01/2024
Ziv Gan-Or and 50 other authors -
Prediction of COVID-19 severity using machine learning – 01/10/2024
Yvan Devaux and 29 other authors -
Sex inequalities in cardiovascular risk factors and their management in primary prevention in adults living with type 1 diabetes in Germany and France – 16/09/2024
Emmanuel Cosson and 9 other authors -
Co-design of a voice-based app to monitor long COVID symptoms with its end-users – 09/09/2024
Aurélie Fischer and 8 other authors -
Can we screen for type 2 diabetes using voice? – 01/09/2024
Guy Fagherazzi and 8 other authors -
Multimodal Fusion for Vocal Biomarkers Using Vector Cross-Attention – 01/09/2024
Vladimir Despotovic and 3 other authors -
The transformative role of artificial intelligence in diabetes care and research – 27/07/2024
Guy Fagherazzi and 4 other authors -
General and abdominal adiposity and hypertension in eight world regions – 31/08/2024
Majid Ezzati and 50 other authors -
Digital Vocal Biomarker of Smoking Status Using Ecological Audio Recordings – 03/07/2024
Guy Fagherazzi and 3 other authors -
Heterogeneity of glycaemic phenotypes in type 1 diabetes – 23/05/2024
Guy Fagherazzi and 29 other authors -
The impact of patient registration on utilisation and quality of care – 12/07/2024
Valerie Moran and 6 other authors -
Cardiovascular history and risk of idiopathic Parkinson’s disease – 08/07/2024
Shubhra Acharya and 47 other authors -
Smartphone use and cerebro-cardio-vascular health – 01/07/2024
Yvan Devaux and 2 other authors -
Assessment of the Impact of Subcutaneous Catheter Change on Glucose Control in Patients with Type 1 Diabetes Treated by Insulin Pump in Open- and Closed-Loop Modes – 01/01/2024
J. P. Riveline and 9 other authors -
A Voice-Based AI Algorithm Can Predict Type 2 Diabetes Status-Findings from the Colive Voice Study on US Adult Participants – 14/06/2024
Abir Elbeji and 8 other authors -
Converging peripheral blood microRNA profiles in Parkinson’s disease and progressive supranuclear palsy – 31/05/2024
Lukas Pavelka and 126 other authors -
Development of a long noncoding RNA-based machine learning model to predict COVID-19 in-hospital mortality – 20/05/2024
Yvan Devaux and 34 other authors -
The use of an automated insulin delivery system is associated with a reduction in diabetes distress and improvement in quality of life in people with type 1 diabetes – 01/01/2024
Yves Reznik and 7 other authors -
Comprehensive blood metabolomics profiling of Parkinson’s disease reveals coordinated alterations in xanthine metabolism – 19/03/2024
Elisa Gómez De Lope and 133 other authors -
Worldwide trends in underweight and obesity from 1990 to 2022 – 01/01/2024
Majid Ezzati and 50 other authors -
ALTRUIST – 05/02/2024
Guy Fagherazzi and 3 other authors -
Who sleeps well in Canada? – 15/11/2023
Saverio Stranges and 10 other authors -
Education as Risk Factor of Mild Cognitive Impairment – 01/01/2024
Matthias Klee and 137 other authors -
Development and validation of a Type 1 and Type 2 diabetes-specific patient-reported experience measure e-questionnaire – 01/01/2024
Coline Hehn and 11 other authors -
Relationship between multimorbidity, functional limitation, and quality of life among middle-aged and older adults – 01/01/2023
Piotr Wilk and 10 other authors -
Author Correction – 18/12/2023
Sinthuja Pachchek and 46 other authors -
The role of multimorbidity and socio-economic characteristics as potential risk factors for Long Covid – 01/12/2023
Piotr Wilk and 6 other authors -
Novel, computational IgE-clustering in a population-based cross-sectional study: Mapping the allergy burden – 01/12/2023
Rebecca Czolk and 10 other authors -
Accurate long-read sequencing identified GBA1 as major risk factor in the Luxembourgish Parkinson’s study – 23/11/2023
Sinthuja Pachchek and 131 other authors -
Construction and validation of the area level deprivation index for health research – 16/11/2023
Ishor Sharma and 7 other authors -
Association between endometriosis and risk of type 2 diabetes – 28/07/2023
Marina Kvaskoff and 5 other authors -
Validation of a SARS-CoV-2 Surrogate Neutralization Test Detecting Neutralizing Antibodies against the Major Variants of Concern – 06/10/2023
Carole Seguin-Devaux and 13 other authors -
Mixed-methods study protocol to identify expectations of people with type 1 diabetes and their caregivers about voice-based digital health solutions to support the management of diabetes distress – 13/09/2023
Gloria Aguayo and 5 other authors -
Catheter changes lead to increased hyperglycaemia in both open and closed loop modes: real-life evidence from pump users with type 1 diabetes – 04/09/2023
Jean-Baptiste Julla and 10 other authors -
Substituting device-measured sedentary time with alternative 24-hour movement behaviours: compositional associations with adiposity, cardiometabolic risk and arterial stiffness in the ORISCAV-LUX 2 study – 01/09/2023
Paul J Collings and 4 other authors -
Novel, computational IgE-clustering in a population-based cross-sectional study – 27/07/2023
Annette Kuehn and 10 other authors -
O.3.2-2 Substituting device-measured sedentary time with alternative 24-hour movement behaviours – 01/09/2023
Paul James Collings and 4 other authors -
The relationship between sleep health and multimorbidity in community dwelling populations – 15/07/2023
P. Nistor and 4 other authors -
The Long COVID experience from a patient’s perspective – 17/08/2023
Guy Fagherazzi and 4 other authors -
Association of diet quality indices with serum and metabolic biomarkers in participants of the ORISCAV-LUX-2 study – 01/01/2023
Farhad Vahid and 28 other authors -
Interaction between plasma phospholipid odd-chain fatty acids and GAD65 autoantibodies on the incidence of adult-onset diabetes – 10/06/2023
Anna Maria Lampousi and 22 other authors -
School nutrition policy and diet quality of children and youth – 28/03/2023
Sisira Sarma and 3 other authors -
An investigation of media reports of digital surveillance within the first year of the COVID-19 pandemic – 24/07/2023
Leigha Comer and 13 other authors -
Technologies will not make diabetes disappear – 01/07/2023
Guy Fagherazzi -
The double burden of malnutrition among women of reproductive age and preschool children in low- and middle-income countries – 31/03/2023
Jason Mulimba Were and 6 other authors -
Co-Design of a Voice-Based Digital Health Solution to Monitor Persisting Symptoms Related to COVID-19 (UpcomingVoice Study) – 19/06/2023
Aurélie Fischer and 6 other authors -
Investigating Health Inequality Using Trend, Decomposition and Spatial Analyses – 02/06/2023
Shehzad Ali and 4 other authors -
Profiles of Glycemic Control in People with Type 1 Diabetes Using CGM—A Clustering Approach in the SFDT1 Study – 01/06/2023
Emmanuel Cosson and 6 other authors -
Does the place of residence influence your risk of being hypertensive? A study-based on Nepal Demographic and Health Survey – 08/03/2023
Ishor Sharma and 6 other authors -
Prevalence and description of skin reactions associated with adhesives in diabetes technology devices in an adult population – 24/05/2023
J. P. Riveline and 14 other authors -
Sleep Problems and Psychological Well-Being – 21/11/2022
Kelly K. Anderson and 5 other authors -
Burden of HIV and treatment outcomes among TB patients in rural Kenya – 30/05/2023
Moses Ngari and 8 other authors -
Do dietary practices and household environmental quality mediate socioeconomic inequalities in child undernutrition risk in West Africa? – 19/10/2022
A Kofi Amegah and 5 other authors -
A nutritional biomarker score of the Mediterranean diet and incident type 2 diabetes – 27/04/2023
Nita G. Forouhi and 33 other authors -
Patients’ and caregivers’ perceptions of bariatric surgery – 18/04/2023
Sébastien Czernichow and 10 other authors -
Substituting device-measured sedentary time with alternative 24-hour movement behaviours – 04/04/2023
Laurent Malisoux and 4 other authors -
Prevalence and Description of the Skin Reactions Associated with Adhesives in Diabetes Technology Devices in an Adult Population – 24/02/2023
J. P. Riveline and 14 other authors -
Association between use of psychotropic medications prior to SARS-COV-2 infection and trajectories of COVID-19 recovery – 16/03/2023
Gloria Aguayo and 5 other authors -
Altered infective competence of the human gut microbiome in COVID-19 – 09/03/2023
Laura de Nies and 28 other authors -
Characterizing the adult exposome in men and women from the general population – 26/01/2023
Maria Ruiz and 8 other authors -
Influence of the Social Environment on Ideal Cardiovascular Health – 21/02/2023
Stephanie J. Frisbee and 4 other authors -
Breakfast Consumption and Diet Quality of Teens in Southwestern Ontario – 01/02/2023
Jason A. Gilliland and 7 other authors -
COVID-19 vaccine inequity and Big Pharma – 15/11/2022
Shehzad Ali and 2 other authors -
Diabetes registries and high-quality diabetes care – 01/02/2023
Kamlesh Khunti and 18 other authors -
Correction – 31/01/2023
Gloria Aguayo and 10 other authors -
Machine learning for predicting neurodegenerative diseases in the general older population – 11/01/2023
Gloria Aguayo and 10 other authors -
PRODIAB – 01/01/2023
Nicolas Naïditch and 6 other authors -
Les réseaux sociaux au service de la recherche sur le diabète – 01/01/2023
Charline Bour and 1 other author -
Strategies to improve health status among adults with multimorbidity – 14/09/2022
Kathryn Nicholson and 4 other authors -
Towards a European “Cohort Moonshot” – 01/01/2023
Guy Fagherazzi -
Associations Between Wearable-Specific Indicators of Physical Activity Behaviour and Insulin Sensitivity and Glycated Haemoglobin in the General Population – 12/12/2022
Anne Backes and 29 other authors -
Nutrition and health – 03/10/2022
Saverio Stranges and 1 other author -
The relationship between sleep and multimorbidity in community dwelling populations: a global perspective – 01/12/2022
P. Nistor and 4 other authors -
Long COVID Classification – 30/11/2022
Aurélie Fischer and 8 other authors -
Identifying Profiles and Symptoms of Patients With Long COVID in France – 22/11/2022
Amélia Déguilhem and 10 other authors -
Vocal biomarker predicts fatigue in people with COVID-19 – 22/11/2022
Guy Fagherazzi and 7 other authors -
Association of miR-144 levels in the peripheral blood with COVID-19 severity and mortality – 21/11/2022
Fabio Martelli and 17 other authors -
Recommendations for Successful Implementation of the Use of Vocal Biomarkers for Remote Monitoring of COVID-19 and Long COVID in Clinical Practice and Research – 15/11/2022
Aurélie Fischer and 3 other authors -
Discovery and Analytical Validation of a Vocal Biomarker to Monitor Anosmia and Ageusia in Patients with COVID-19 – 07/09/2022
Guy Fagherazzi and 6 other authors -
A voice-based biomarker for monitoring symptom resolution in adults with COVID-19 – 20/10/2022
Guy Fagherazzi and 8 other authors -
Global diabetes burden – 01/10/2022
Guy Fagherazzi and 5 other authors -
Association between functional limitation and quality of life among older adults with multimorbidity in Luxembourg – 01/10/2022
Piotr Wilk and 9 other authors -
How multimorbidity and socio-economic factors affect Long COVID: Evidence from European Countries – 01/10/2022
Piotr Wilk and 6 other authors -
Analysing breast cancer survivors’ acceptance profiles for using an electronic pillbox connected to a smartphone application using Seintinelles, a French community-based research tool – 27/09/2022
Catherine Goetzinger and 6 other authors -
Publisher Correction – 02/09/2022
Lukas Pavelka and 47 other authors -
Association between endometriosis and risk of type 2 diabetes: results of the E3N prospective cohort study – 01/09/2022
P. Vaduva and 4 other authors -
Associations between Major Health Behaviors and Sleep Problems – 01/01/2021
Piotr Wilk and 4 other authors -
Associations between sleep patterns, smoking, and alcohol use among older adults in Canada – 01/09/2022
Kelly K. Anderson and 6 other authors -
Precision Health – 01/09/2022
Guy Fagherazzi and 11 other authors -
Precision Health – 01/09/2022
Guy Fagherazzi and 11 other authors -
Precision Health – 01/09/2022
Guy Fagherazzi and 11 other authors -
Residential greenness and substance use among youth and young adults – 24/03/2022
Jamie A. Seabrook and 4 other authors -
Secular trends in low birth weight and child undernutrition in West Africa – 01/09/2022
A Kofi Amegah and 6 other authors -
Does Shiftwork Impact Cognitive Performance? Findings from the Canadian Longitudinal Study on Aging (CLSA) – 16/08/2022
Saverio Stranges and 6 other authors -
Age at onset as stratifier in idiopathic Parkinson’s disease – effect of ageing and polygenic risk score on clinical phenotypes – 09/08/2022
Lukas Pavelka and 50 other authors -
Long COVID Symptomatology After 12 Months and Its Impact on Quality of Life According to Initial Coronavirus Disease 2019 Disease Severity – 05/08/2022
Guy Fagherazzi and 8 other authors -
Examining the prevalence and correlates of multimorbidity among community-dwelling older adults – 02/08/2022
Kathryn Nicholson and 5 other authors -
An individualized functional magnetic resonance imaging protocol to assess semantic congruency effects on episodic memory in an aging multilingual population – 22/07/2022
Magali Perquin and 8 other authors -
Extraction of Explicit and Implicit Cause-Effect Relationships in Patient-Reported Diabetes-Related Tweets From 2017 to 2021 – 19/07/2022
Adrian Ahne and 8 other authors -
Emulating a virtual digital cohort study based on social media data as a complementary approach to traditional epidemiology – 24/05/2022
Guy Fagherazzi and 2 other authors -
The Nutri-Score nutrition label – 27/07/2021
Serge Hercberg and 6 other authors -
Dynamics of SARS-CoV-2 spreading under the influence of environmental factors and strategies to tackle the pandemic – 01/06/2022
Zunaira Asif and 8 other authors -
An individualized fMRI-based protocol to assess semantic memory in a heterogeneous multilingual population – 22/05/2022
Magali Perquin and 8 other authors -
Association of neighbourhood disadvantage and individual socioeconomic position with all-cause mortality – 01/05/2022
Ana Isabel Ribeiro and 49 other authors -
Prevalence and Cost of Care for Parkinson’s Disease in Luxembourg – 01/01/2022
Michel Vaillant and 7 other authors -
Small steps, strong shield – 01/05/2022
Jon Patricios and 9 other authors -
Associations between physical activity prior to infection and COVID-19 disease severity and symptoms – 29/04/2022
Laurent Malisoux and 5 other authors -
Impact of intra-category food substitutions on the risk of type 2 diabetes – 01/01/2021
Moufidath Adjibade and 7 other authors -
Identification of vocal biomarkers for screening diabetes and monitoring health of people with diabetes: preliminary results from the COLIVE voice study – 25/04/2022
Abir Elbeji and 3 other authors -
Combinatorial analysis reveals highly coordinated early-stage immune reactions that predict later antiviral immunity in mild COVID-19 patients – 01/01/2022
Christophe Capelle and 23 other authors -
Interim analysis of COVID-19 vaccine effectiveness against Severe Acute Respiratory Infection due to laboratory-confirmed SARS-CoV-2 among individuals aged 30 years and older, ECDC multi-country study – second update – 14/03/2022
Nassera Aouali and 4 other authors -
Attitudes and Expectations of Clinical Research Participants Toward Digital Health and Mobile Dietary Assessment Tools – 09/03/2022
Florent Schäfer and 6 other authors -
Associations Between Device-Measured Physical Activity and Glycemic Control and Variability Indices Under Free-Living Conditions – 14/12/2021
Douae El Fatouhi and 7 other authors -
Comparison of Deep Neural Networks and Regularised Cox Regression Models in the Prediction of Neurodegenerative Diseases in the General Older Population – 01/03/2022
Gloria Aguayo and 10 other authors -
The prevalence of physical multimorbidity among people with non-affective psychotic disorders 10 years after first diagnosis – 01/01/2021
Kelly K. Anderson and 3 other authors -
Examining Variations in the Prevalence of Diagnosed Mood or Anxiety Disorders Among Migrant Groups in Ontario, 1995-2015 – 01/02/2022
Jordan Edwards and 5 other authors -
Global Vaccine Equity to End the COVID-19 Pandemic – 27/01/2022
Michael Clarke and 3 other authors -
Association between Dietary Factors and Constipation in Adults Living in Luxembourg and Taking Part in the ORISCAV-LUX 2 Survey – 28/12/2021
Maurane Rollet and 17 other authors -
Advanced analytical methods to assess physical activity behavior using accelerometer time series – 01/01/2021
Anne Backes and 4 other authors -
Improving Diabetes-Related Biomedical Literature Exploration in the Clinical Decision-making Process via Interactive Classification and Topic Discovery – 01/01/2022
Adrian Ahne and 4 other authors -
Objective and subjective sleep measures are associated with HbA1c and insulin sensitivity in the general population – 20/05/2021
Gloria Aguayo and 10 other authors -
Secular trends of ischaemic heart disease, stroke, and dementia in high-income countries from 1990 to 2017 – 01/01/2021
Negar Morovatdar and 11 other authors -
Cardiometabolic Health Associated with Exposures to Mixtures of Chemical Pollutants: Results from the EHES-LUX Study – 01/01/2022
Giovana M. Ciprián and 6 other authors -
Long COVID Classification: Findings from a Clustering Analysis in the Predi-COVID Cohort Study – 01/01/2022
Aurélie Fischer and 8 other authors -
Multimorbidity in large Canadian urban centres: A multilevel analysis of pooled 2015–2018 cross-sectional cycles of the Canadian Community Health Survey – 20/12/2021
Piotr Wilk and 7 other authors -
Examining the double burden of malnutrition for preschool children and women of reproductive age in low-income and middle-income countries – 14/12/2021
Jason Mulimba Were and 4 other authors -
Clustering of obesity-related characteristics – 01/12/2021
Laura N. Anderson and 8 other authors -
Estimated visceral adiposity is associated with risk of cardiometabolic conditions in a population based study – 01/12/2021
Maria Ruiz and 5 other authors -
Methods to Generate Innovative Research Ideas and Improve Patient and Public Involvement in Modern Epidemiological Research – 01/12/2021
Gloria Aguayo and 9 other authors -
Allergy burden in luxembourg: A population-based, cross-sectional health survey paralleled by component-resolved-IgE-diagnosis – 15/11/2021
Rebecca Czolk and 13 other authors -
Identifying causal associations in tweets using deep learning: Use case on diabetes-related tweets from 2017-2021 – 01/11/2021
Adrian Ahne and 8 other authors -
Mapping the human chemical exposome for public health – 18/03/2026
Arthur David and 57 other authors -
Pesticide Contamination in the Hair of Children From Colonia San Juan, a Rural Community in Paraguay – 26/12/2025
Stela Benitez Leite and 5 other authors -
27th Meeting of the Society of Hair Testing (SoHT) – 12/12/2025
Donata Favretto and 3 other authors -
Chemical pollutant mixtures associated with metabolic health – 10/10/2025
Jesús Martínez Gómez and 6 other authors -
Polycyclic aromatic hydrocarbon (PAH) exposure among European adults – 16/03/2025
Dimosthenis Sarigiannis and 31 other authors -
Exposure of wild mammals to glyphosate, AMPA, and glufosinate – 23/01/2025
Clémentine Fritsch and 11 other authors -
Comprehensive analysis, comprehensive understanding – 17/12/2024
Brice Appenzeller and 1 other author -
Cardiovascular risk factors in relation to hair polycyclic aromatic hydrocarbons in the NESCAV study – 27/11/2024
Brice Appenzeller and 9 other authors -
Exposure assessment of the European adult population to deoxynivalenol – Results from the HBM4EU Aligned Studies – 01/12/2024
Sónia Namorado and 24 other authors -
Cross-Sectional Examination of Thyroid Hormones and Environmental Exposure to Multiclass Pesticides in Women of Reproductive Age in China – 18/10/2024
Brice Appenzeller and 6 other authors -
Exposure to pesticides, persistent and non − persistent pollutants in French 3.5-year-old children – 07/07/2024
Florence Zeman and 5 other authors -
PBPK modeling to support risk assessment of pyrethroid exposure in French pregnant women – 08/03/2024
Florence Zeman and 7 other authors -
Glucocorticoid hormones in relation to environmental exposure to bisphenols and multiclass pesticides among middle aged-women – 22/03/2024
Brice Appenzeller and 6 other authors -
Hair-Derived Exposome Exploration of Cardiometabolic Health – 24/03/2024
Marc Chadeau-Hyam and 13 other authors -
A meta-analysis highlighting the increasing relevance of the hair matrix in exposure assessment to organic pollutants – 01/02/2024
Muhammad Junaid and 7 other authors -
Publisher Correction – 29/01/2024
Federica Laguzzi and 35 other authors -
Association of hair polychlorinated biphenyls and multiclass pesticides with obesity, diabetes, hypertension and dyslipidemia in NESCAV study – 27/09/2023
Brice Appenzeller and 10 other authors -
Association between Environmental Exposure to Multiclass Organic Pollutants and Sex Steroid Hormone Levels in Women of Reproductive Age – 07/11/2023
Brice Appenzeller and 6 other authors -
Determinants of exposure to acrylamide in European children and adults based on urinary biomarkers – 02/12/2023
Laguzzi Federica and 35 other authors -
Time Patterns in Internal Human Exposure Data to Bisphenols, Phthalates, DINCH, Organophosphate Flame Retardants, Cadmium and Polyaromatic Hydrocarbons in Europe – 28/09/2023
Laura Rodriguez Martin and 61 other authors -
Chemical Exposure Highlighted without Any A Priori Information in an Epidemiological Study by Metabolomic FT-ICR-MS Fingerprinting at High Throughput and High Resolution – 28/02/2023
Estelle Rathahao-Paris and 8 other authors -
Advancing in hair testing – 01/01/2023
Donata Favretto and 3 other authors -
Environmental tobacco smoke at home and in public places prior to smoking ban enforcement – 23/12/2022
Brice Appenzeller and 5 other authors -
The Society of Hair Testing consensus on general recommendations for hair testing and drugs of abuse testing in hair – 01/01/2023
Donata Favretto and 14 other authors -
Association of diet quality indices with serum and metabolic biomarkers in participants of the ORISCAV-LUX-2 study – 01/01/2023
Farhad Vahid and 28 other authors -
Transcriptomic profiling reveals differential cellular response to copper oxide nanoparticles and polystyrene nanoplastics in perfused human placenta – 07/06/2023
Tina Buerki-Thurnherr and 10 other authors -
Skin microbiome differentiates into distinct cutotypes with unique metabolic functions upon exposure to polycyclic aromatic hydrocarbons – 01/06/2023
Patrick K.H. Lee and 13 other authors -
Incorporation of Fast-Elimination Chemicals in Hair Is Governed by Pharmacokinetics-Implications for Exposure Assessment – 01/01/2022
Brice Appenzeller and 7 other authors -
Harmonized human biomonitoring in European children, teenagers and adults – 09/02/2023
Eva Govarts and 110 other authors -
Characterizing the adult exposome in men and women from the general population – 26/01/2023
Maria Ruiz and 8 other authors -
Skin microbiome differentiates into distinct cutotypes with unique metabolic functions upon exposure to polycyclic aromatic hydrocarbons – 09/02/2023
Patrick K.H. Lee and 10 other authors -
Associations Between Wearable-Specific Indicators of Physical Activity Behaviour and Insulin Sensitivity and Glycated Haemoglobin in the General Population – 12/12/2022
Anne Backes and 29 other authors -
Measurement of hair thyroid and steroid hormone concentrations in the rat evidence endocrine disrupting potential of a low dose mixture of polycyclic aromatic hydrocarbons – 15/09/2022
Brice Appenzeller and 3 other authors -
Pervasive exposure of wild small mammals to legacy and currently used pesticide mixtures in arable landscapes – 23/09/2022
Clémentine Fritsch and 18 other authors -
Cadmium exposure in adults across Europe – 01/09/2022
Janja Snoj Tratnik and 26 other authors -
Comprehensive Assessment of Local Population Chemical Exposome by Combination of Organic Pollutant- and Metal-Multi-Residue Analysis in Hair – 01/01/2021
Alba Iglesias Gonzalez and 6 other authors -
Hormonal profile changes induced by pesticide mixture exposure in female rats revealed by hair analysis – 26/05/2022
Brice Appenzeller and 3 other authors -
Precision Health – 01/09/2022
Guy Fagherazzi and 11 other authors -
Precision Health – 01/09/2022
Guy Fagherazzi and 11 other authors -
Precision Health – 01/09/2022
Guy Fagherazzi and 11 other authors -
Time Trends of Acrylamide Exposure in Europe – 17/08/2022
Michael Poteser and 28 other authors -
Trends of Exposure to Acrylamide as Measured by Urinary Biomarkers Levels within the HBM4EU Biomonitoring Aligned Studies (2000–2021) – 02/08/2022
Michael Poteser and 29 other authors -
Biomonitoring of children exposure to pollutants based on hair analysis – 13/07/2022
Alba Iglesias Gonzalez -
Investigating children’s chemical exposome – Description and possible determinants of exposure in the region of Luxembourg based on hair analysis – 11/06/2022
Alba Iglesias Gonzalez and 6 other authors -
Harmonization of Human Biomonitoring Studies in Europe – 01/06/2022
Liese Gilles and 60 other authors -
Differences and Interactions in Placental Manganese and Iron Transfer across an In Vitro Model of Human Villous Trophoblasts – 18/03/2022
Julia Bornhorst and 7 other authors -
Profiling steroid and thyroid hormones with hair analysis in a cohort of women aged 25 to 45 years old – 18/03/2022
Brice Appenzeller and 5 other authors -
Chimpanzee exposure to pollution revealed by human biomonitoring approaches – 15/03/2022
Sabrina Krief and 8 other authors -
Association between Dietary Factors and Constipation in Adults Living in Luxembourg and Taking Part in the ORISCAV-LUX 2 Survey – 28/12/2021
Maurane Rollet and 17 other authors -
Cardiometabolic Health Associated with Exposures to Mixtures of Chemical Pollutants: Results from the EHES-LUX Study – 01/01/2022
Giovana M. Ciprián and 6 other authors -
Linking Macroinvertebrates and Physicochemical Parameters for Water Quality Assessment in the Lower Basin of the Volta River in Ghana – 01/01/2021
Paul J. Van den Brink and 3 other authors -
(Eco)toxicological tests for assessing impacts of chemical stress to aquatic ecosystems – 15/11/2021
Lara M. Schuijt and 4 other authors -
Nexus between perfluoroalkyl compounds (PFCs) and human thyroid dysfunction – 01/01/2020
Weiping Xie and 5 other authors -
Multi-omics analysis to decipher the molecular link between chronic exposure to pollution and human skin dysfunction – 15/09/2021
Namita Misra and 13 other authors -
Population-based biomonitoring of exposure to persistent and non-persistent organic pollutants in the Grand Duchy of Luxembourg – 01/08/2021
Brice Appenzeller and 6 other authors -
Biomonitoring of fast-elimination endocrine disruptors – Results from a 6-month follow up on human volunteers with repeated urine and hair collection – 15/07/2021
Brice Appenzeller and 5 other authors -
Novel Strategies for the assessment of exposure to fast elimination endocrine disruptors with non-invasive biological sampling – 13/07/2021
François Fays -
Hair versus urine for the biomonitoring of pesticide exposure – 01/07/2021
Brice Appenzeller and 7 other authors -
Impact of protein-enriched plant food items on the bioaccessibility and cellular uptake of carotenoids – 23/06/2021
Torsten Bohn and 7 other authors -
Double constrained ordination for assessing biological trait responses to multiple stressors – 01/02/2021
Fengjiao Peng and 3 other authors -
Micronutrients and markers of oxidative stress and inflammation related to cardiometabolic health – 22/12/2020
Maria Ruiz and 8 other authors -
Is there an optimal sampling time and number of samples for assessing exposure to fast elimination endocrine disruptors with urinary biomarkers? – 10/12/2020
François Fays and 10 other authors -
Human exposure to PCBs, PBDEs and bisphenols revealed by hair analysis – 01/12/2020
Fengjiao Peng and 11 other authors -
Road impact in a protected area with rich biodiversity – 01/08/2020
Sabrina Krief and 9 other authors -
Skin exposome science in practice – 01/07/2020
L. Aguilar and 2 other authors -
Changes of the human skin microbiota upon chronic exposure to polycyclic aromatic hydrocarbon pollutants – 26/06/2020
Patrick K.H. Lee and 12 other authors -
Erratum – 02/06/2020
Artem Krasnobaev and 7 other authors -
Exposure to multiclass pesticides among female adult population in two Chinese cities revealed by hair analysis – 01/05/2020
Brice Appenzeller and 9 other authors -
Benthic invertebrate and microbial biodiversity in sub-tropical urban rivers – 20/03/2020
Fengjiao Peng and 7 other authors -
Legacy and Emerging Persistent Organic Pollutants in Antarctic Benthic Invertebrates near Rothera Point, Western Antarctic Peninsula – 03/03/2020
Nico W. Van Den Brink and 7 other authors -
Alternative halogenated flame retardants (AHFRs) in green mussels from the south China sea – 01/03/2020
Runxia Sun and 5 other authors -
Ultra performance liquid chromatography – tandem mass spectrometer method applied to the analysis of both thyroid and steroid hormones in human hair – 08/02/2020
Nathalie Grova and 5 other authors -
Occurrence and congener profiles of polybrominated diphenyl ethers in green mussels (Perna viridis) collected from northern South China Sea and the associated potential health risk – 01/01/2020
Runxia Sun and 4 other authors -
Cumulative exposure to organic pollutants of French children assessed by hair analysis – 01/01/2020
Alba Iglesias Gonzalez and 2 other authors -
Multiple pesticides in mothers’ hair samples and children’s measurements at birth – 01/01/2020
Rémi Béranger and 6 other authors -
PAHs increase the production of extracellular vesicles both in vitro in endothelial cells and in vivo in urines from rats – 01/12/2019
Eric Le Ferrec and 11 other authors -
Blood pharmacokinetic of 17 common pesticides in mixture following a single oral exposure in rats – 01/10/2019
Caroline Chata and 6 other authors -
Polycyclic Aromatic Hydrocarbons Can Trigger Hepatocyte Release of Extracellular Vesicles by Various Mechanisms of Action Depending on Their Affinity for the Aryl Hydrocarbon Receptor – 01/10/2019
Odile Sergent and 13 other authors -
Human pollution exposure correlates with accelerated ultrastructural degradation of hair fibers – 10/09/2019
Thomas Bornschlögl and 7 other authors -
Biomonitoring of children exposure to urban pollution and environmental tobacco smoke with hair analysis – A pilot study on children living in Paris and Yeu Island, France – 15/05/2019
Brice Appenzeller and 2 other authors -
Challenges and benefits of integrating diverse sampling strategies in the observation of cardiovascular risk factors (ORISCAV-LUX 2) study – 04/02/2019
Alaa Al Kerwi and 17 other authors -
Exposure to Polycyclic Aromatic Hydrocarbons Leads to Non-monotonic Modulation of DNA and RNA (hydroxy)methylation in a Rat Model – 01/12/2018
Radu Duca and 7 other authors -
Exposure to polycyclic aromatic hydrocarbons in women living in the Chinese cities of BaoDing and Dalian revealed by hair analysis – 01/12/2018
Brice Appenzeller and 9 other authors -
Mechanisms involved in the death of steatotic WIF-B9 hepatocytes co-exposed to benzo[a]pyrene and ethanol – 01/12/2018
Dominique Lagadic-Gossmann and 13 other authors -
Regulatory identification of BPA as an endocrine disruptor – 05/11/2018
C. Beausoleil and 27 other authors -
Multiple pesticide analysis in hair samples of pregnant French women – 01/11/2018
Rémi Béranger and 8 other authors -
Exposure to environmental levels of polycyclic aromatic hydrocarbons leads to epigenetic modulation in a rat model – 10/10/2018
Radu Duca and 6 other authors -
Hair analysis for the biomonitoring of polycyclic aromatic hydrocarbon exposure – 01/10/2018
Nathalie Grova and 4 other authors -
Identification of beta-arrestin-1 as a diagnostic biomarker in lung cancer – 28/08/2018
Victoria El Khoury and 13 other authors -
Study of the incorporation mechanism of organic chemicals into hair – 29/05/2018
Caroline Chata -
Potential of dynamically harmonized Fourier transform ion cyclotron resonance cell for high-throughput metabolomics fingerprinting – 01/01/2018
Estelle Rathahao-Paris and 6 other authors -
Identification of new tetrahydroxylated metabolites of Polycyclic Aromatic Hydrocarbons in hair as biomarkers of exposure and signature of DNA adduct levels – 01/12/2017
Nathalie Grova and 6 other authors -
Chemical evaluation of electronic cigarettes – 01/10/2017
Anne Garat and 13 other authors -
Microrna 150-5p improves risk classification for mortality within 90 days after acute ischemic stroke – 01/09/2017
Mira Katan and 7 other authors -
Hair analysis for the biomonitoring of pesticide exposure – 01/08/2017
Brice Appenzeller and 7 other authors -
Protein S100 as outcome predictor after out-of-hospital cardiac arrest and targeted temperature management at 33 °C and 36 °C – 20/06/2017
Pascal Stammet and 21 other authors -
Genetic and epigenetic alterations in normal and sensitive COPD-diseased human bronchial epithelial cells repeatedly exposed to air pollution-derived PM2.5 – 01/01/2017
G. Garçon and 13 other authors -
New insights into urine-based assessment of polycyclic aromatic hydrocarbon-exposure from a rat model – 01/01/2017
Nathalie Grova and 3 other authors -
Role for the ATPase inhibitory factor 1 in the environmental carcinogen-induced Warburg phenotype – 01/01/2017
Dominique Lagadic-Gossmann and 13 other authors -
Reduced rapid eye movement density in Parkinson disease – 01/12/2016
Nico J. Diederich and 5 other authors -
Differential responses of healthy and chronic obstructive pulmonary diseased human bronchial epithelial cells repeatedly exposed to air pollution-derived PM4 – 01/11/2016
G. Garçon and 10 other authors -
Mortality prediction of 35 frailty scores in a 7-years follow-up study in elderly general population – 01/11/2016
Gloria Aguayo and 8 other authors -
Determination of tetrahydroxylated metabolites in hair and DNA of rats under controlled exposure to a mixture of Polycyclic Aromatic Hydrocarbons: new biomarkers for assessing human exposure – 01/09/2016
Nathalie Grova and 3 other authors -
Epigenetic changes upon multi-residue exposure to polycyclic aromatic hydrocarbons – 01/09/2016
Radu Duca and 5 other authors -
Influence of pesticide physicochemical properties on the association between plasma and hair concentration – 01/05/2016
Brice Appenzeller and 3 other authors -
Analysis of tetrahydroxylated benzo[a]pyrene isomers in hair as biomarkers of exposure to benzo[a]pyrene – 01/03/2016
Nathalie Grova and 3 other authors -
Behavioral toxicity and physiological changes from repeated exposure to fluorene administered orally or intraperitoneally to adult male Wistar rats – 01/03/2016
Julie Peiffer and 7 other authors -
Pesticide detection in air samples from contrasted houses and in their inhabitants’ hair – 15/02/2016
Caroline Raeppel and 3 other authors -
Multi-residue analysis of organic pollutants in hair and urine for matrices comparison – 01/04/2015
Brice Appenzeller and 3 other authors -
Analysis of airborne pesticides from different chemical classes adsorbed on Radiello® Tenax® passive tubes by thermal-desorption-GC/MS – 01/02/2015
Maurice Millet and 6 other authors -
Hair Analysis for the Biomonitoring of Human Exposure to Organic Pollutants – 01/01/2015
Brice Appenzeller -
Determination of seven pyrethroids biocides and their synergist in indoor air by thermal-desorption gas chromatography/mass spectrometry after sampling on Tenax TA® passive tubes – 01/01/2015
Maurice Millet and 2 other authors -
Tetrahydroxylated-benzo[a]pyrene isomer analysis after hydrolysis of DNA-adducts isolated from rat and human white blood cells – 17/10/2014
Nathalie Grova and 4 other authors -
Short-term effects of a perinatal exposure to a 16 polycyclic aromatic hydrocarbon mixture in rats – 01/07/2014
Guillemette Crépeaux and 8 other authors -
Hair decontamination procedure prior to multi-class pesticide analysis – 01/06/2014
Brice Appenzeller and 3 other authors -
Comparison of solid phase- and liquid/liquid-extraction for the purification of hair extract prior to multi-class pesticides analysis – 01/04/2014
Brice Appenzeller and 3 other authors -
Coupling ASE, sylilation and SPME-GC/MS for the analysis of current-used pesticides in atmosphere – 01/04/2014
Maurice Millet and 4 other authors -
An update of issues regarding hair analysis of organic pollutants – 01/02/2014
M. Kavvalakis and 2 other authors -
Monitoring of polycyclic aromatic hydrocarbons (PAHs) in the atmosphere of southern Luxembourg using XAD-2 resin-based passive samplers – 01/02/2014
Maurice Millet and 2 other authors -
Gas chromatography-tandem mass spectrometry analysis of 52 monohydroxylated metabolites of polycyclic aromatic hydrocarbons in hairs of rats after controlled exposure – 01/11/2013
Nathalie Grova and 2 other authors -
Neurobehavioral Toxicity of a Repeated Exposure (14 Days) to the Airborne Polycyclic Aromatic Hydrocarbon Fluorene in Adult Wistar Male Rats – 20/08/2013
Henri Schroeder and 9 other authors -
Application of XAD-2 resin-based passive samplers and SPME-GC-MS/MS analysis for the monitoring of spatial and temporal variations of atmospheric pesticides in Luxembourg – 01/11/2012
Maurice Millet and 4 other authors -
Determination of farm workers’ exposure to pesticides by hair analysis – 25/04/2012
Brice Appenzeller and 4 other authors -
Hair analysis for biomonitoring of environmental and occupational exposure to organic pollutants – 25/04/2012
Brice Appenzeller and 1 other author -
Reply to – 25/04/2012
Brice Appenzeller and 1 other author -
Simultaneous determination of nicotine and PAH metabolites in human hair specimen – 25/04/2012
Brice Appenzeller and 4 other authors -
Multi-class pesticide analysis in human hair by gas chromatography tandem (triple quadrupole) mass spectrometry with solid phase microextraction and liquid injection – 13/01/2012
Brice Appenzeller and 4 other authors -
Simultaneous analysis of pesticides from different chemical classes by using a derivatisation step and gas chromatography-mass spectrometry – 04/11/2011
Maurice Millet and 5 other authors -
Impact of hair-care products on FAEE hair concentrations in substance abuse monitoring – 01/04/2011
Joey Gareri and 3 other authors -
1,5-Electrocyc-lization of 1-alkyl-3-[(2Z)-2,4-diaryl-4-oxobut-2-en-1-yl]- 1H-benzimidazol-3-ium bromides – 01/01/2011
L. M. Potikha and 2 other authors -
Synthesis of azepino[1,2-a]benzimidazole and imidazo[1,2-a]azepine derivatives – 01/01/2011
L. M. Potikha and 2 other authors -
Internet Suicide Guidelines – 01/06/2010
Serge Schneider and 5 other authors -
Temporal variations of concentrations of currently used pesticides in the atmosphere of Strasbourg, France – 01/02/2010
Maurice Millet and 5 other authors -
A new method or pyridine cycle attachment to the derivatives of imidazole and benzimidazole – 01/01/2010
L. M. Potikha and 3 other authors -
An interaction of (Z)-4-bromo-1,3-di(2-thieny1)-2-buten-1-one with amines, the synthesis of di(2-thieny1)azolo[a]pyridines – 01/01/2010
L. M. Potikha and 6 other authors -
Gas/particle partitioning of currently used pesticides in the atmosphere of Strasbourg (France) – 01/01/2010
Maurice Millet and 4 other authors -
Synthesis of diarylazolo[a]pyridines from [(Z)-2,4-diaryl-4-oxo-2-butenyl] azolium salts – 01/01/2010
L. M. Potikha and 3 other authors -
Determination of hydroxylated metabolites of polycyclic aromatic hydrocarbons in human hair by gas chromatography-negative chemical ionization mass spectrometry – 07/08/2009
Claude Schummer and 3 other authors -
Comparison of MTBSTFA and BSTFA in derivatization reactions of polar compounds prior to GC/MS analysis – 15/02/2009
Claude Schummer and 4 other authors -
Synthesis and properties of Z-1,3-bis(aryI)-4-bromo-2-buten-1-ones – 01/01/2009
L. M. Potikha and 2 other authors -
Quantitative determination of ethyl glucuronide in sweat – 01/08/2008
Claude Schummer and 2 other authors -
Determination of fentanyl in sweat and hair of a patient using transdermal patches – 01/04/2008
Serge Schneider and 6 other authors -
Synthesis of 11-methyl-7,9-diphenyl-6H,11H-azepino[2,1-b]benzimidazol-5-ium bromide – 01/01/2008
V. A. Kovtunenko and 3 other authors -
Segmental determination of ethyl glucuronide in hair – 20/12/2007
Brice Appenzeller and 4 other authors -
Determination of the volume of sweat accumulated in a sweat-patch using sodium and potassium as internal reference – 01/06/2007
Brice Appenzeller and 3 other authors -
Ethyl glucuronide concentration in hair is not influenced by pigmentation – 01/01/2007
Brice Appenzeller and 3 other authors -
Drugs and chronic alcohol abuse in drivers – 20/12/2005
Brice Appenzeller and 4 other authors -
Altered distribution of transferrin isoforms according to serum storage conditions – 01/11/2005
Brice Appenzeller and 1 other author -
Relationship between blood alcohol concentration and carbohydrate-deficient transferrin among drivers – 01/08/2005
Brice Appenzeller and 3 other authors -
Reliability of a wireless instrumented insole (WalkinSense system) for measuring spatiotemporal and kinematic gait variables – 22/01/2026
Melanie Eckelt and 9 other authors -
Physical activity in carotid atherosclerotic patients blunts monocyte pro-inflammatory processes – 01/01/2026
Vincent Pialoux and 12 other authors -
An app-based physical activity intervention for people with hip and knee osteoarthritis – 05/12/2025
Laurent Malisoux and 4 other authors -
Association of Shoe Cushioning Perception and Comfort With Injury Risk in Leisure-Time Runners – 22/10/2025
Laurent Malisoux and 4 other authors -
Development and validation of a new simulator of running impacts – 20/08/2025
Robin Gassier and 3 other authors -
Validity of a wireless instrumented insole (WalkinSense system) for measuring gait metrics – 27/09/2025
Melanie Eckelt and 10 other authors -
Geographic environments, daily activities and stress in Luxembourg (the FragMent study) – 02/09/2025
Camille Perchoux and 14 other authors -
Duty Factor Dominates Stride Frequency to Modify Musculoskeletal Peak Loading in Running – 01/08/2025
Fiers Pieter and 6 other authors -
Influence of rear- and fore-part midsole stiffness on running metabolic power – 20/06/2025
Cedric Y. M. Morio and 3 other authors -
Digital twin systems for musculoskeletal applications – 01/01/2025
Pedro Diniz and 8 other authors -
Neighbourhood exposure to fast-food and sit-down restaurants and estimated 24-hour urinary sodium excretion – 01/01/2025
Camille Perchoux and 6 other authors -
National, regional, and global trends in insufficient physical activity among adults from 2000 to 2022 – 01/08/2024
Tessa Strain and 49 other authors -
‘Let me recommend… ‘ – 31/07/2024
Sarah Forberger and 9 other authors -
Associations of movement behaviours and dietary intake with arterial stiffness – 27/07/2024
Laurent Malisoux and 6 other authors -
Longitudinal study of changes in greenness exposure, physical activity and sedentary behavior in the ORISCAV-LUX cohort study – 21/05/2024
Juliette F.E. van Beek and 8 other authors -
Local retail food environment exposure and diet quality in rural and urban adults – 01/05/2024
Marion Tharrey and 8 other authors -
Multicomponent (bio)markers for obesity risk prediction – 08/03/2024
Torsten Bohn and 21 other authors -
Running-Related Injuries Among More Than 7000 Runners in 87 Different Countries – 16/11/2023
Rasmus Oestergaard Nielsen and 3 other authors -
Gait asymmetry in spatiotemporal and kinetic variables does not increase running-related injury risk in lower limbs – 07/12/2023
Laurent Malisoux and 4 other authors -
Ten-Year Changes in the Physical Activity and Sedentary Behaviors of Adults – 21/08/2023
Marina Christofoletti and 4 other authors -
Reference Values and Determinants of Spatiotemporal and Kinetic Variables in Recreational Runners – 19/10/2023
Laurent Malisoux and 4 other authors -
Device-measured physical activity and sedentary time in a national sample of Luxembourg residents: moving beyond the traditional metrics in the ORISCAV-LUX 2 study – 01/09/2023
Laurent Malisoux and 3 other authors -
Longitudinal associations between residential greenness exposure, physical activity and sedentary behavior levels: a country-wide study in Luxembourg – 01/09/2023
J. E.A. van Beek and 7 other authors -
Substituting device-measured sedentary time with alternative 24-hour movement behaviours: compositional associations with adiposity, cardiometabolic risk and arterial stiffness in the ORISCAV-LUX 2 study – 01/09/2023
Paul J Collings and 4 other authors -
O.3.2-2 Substituting device-measured sedentary time with alternative 24-hour movement behaviours – 01/09/2023
Paul James Collings and 4 other authors -
Arterial stiffness and the reallocation of time between device-measured 24-hour movement behaviours – 05/07/2023
Laurent Malisoux and 2 other authors -
Association of diet quality indices with serum and metabolic biomarkers in participants of the ORISCAV-LUX-2 study – 01/01/2023
Farhad Vahid and 28 other authors -
Urban densification over 9 years and change in the metabolic syndrome – 08/06/2023
Marion Tharrey and 4 other authors -
Nine-year exposure to residential greenness and the risk of metabolic syndrome among Luxembourgish adults – 05/04/2023
Marion Tharrey and 4 other authors -
Nine-year exposure to residential greenness and the risk of metabolic syndrome among Luxembourgish adults – 27/04/2023
Marion Tharrey and 4 other authors -
Substituting device-measured sedentary time with alternative 24-hour movement behaviours – 04/04/2023
Laurent Malisoux and 4 other authors -
Status and Trends of Physical Activity Surveillance, Policy, and Research in 164 Countries – 19/12/2022
Andrea Ramírez Varela and 165 other authors -
Lower impact forces but greater burden for the musculoskeletal system in running shoes with greater cushioning stiffness – 01/01/2022
Laurent Malisoux and 4 other authors -
Associations Between Wearable-Specific Indicators of Physical Activity Behaviour and Insulin Sensitivity and Glycated Haemoglobin in the General Population – 12/12/2022
Anne Backes and 29 other authors -
Device-measured physical activity and sedentary time in a national sample of Luxembourg residents – 01/12/2022
Laurent Malisoux and 3 other authors -
Sports injury prevention programmes from the sports physical therapist’s perspective – 07/04/2022
Luciana D. Mendonça and 28 other authors -
Associations between physical activity prior to infection and COVID-19 disease severity and symptoms – 29/04/2022
Laurent Malisoux and 5 other authors -
Associations Between Device-Measured Physical Activity and Glycemic Control and Variability Indices Under Free-Living Conditions – 14/12/2021
Douae El Fatouhi and 7 other authors -
Spatiotemporal and Ground-Reaction Force Characteristics as Risk Factors for Running-Related Injury – 20/01/2022
Laurent Malisoux and 4 other authors -
Association between Dietary Factors and Constipation in Adults Living in Luxembourg and Taking Part in the ORISCAV-LUX 2 Survey – 28/12/2021
Maurane Rollet and 17 other authors -
Advanced analytical methods to assess physical activity behavior using accelerometer time series – 01/01/2021
Anne Backes and 4 other authors -
Objective and subjective sleep measures are associated with HbA1c and insulin sensitivity in the general population – 20/05/2021
Gloria Aguayo and 10 other authors -
Relevance of Frequency-Domain Analyses to Relate Shoe Cushioning, Ground Impact Forces and Running Injury Risk – 11/11/2021
Laurent Malisoux and 5 other authors -
Towards precision cardiometabolic prevention – 06/08/2021
Guy Fagherazzi and 10 other authors -
The effect of shoe cushioning on injury risk, landing impact forces and spatiotemporal parameters during running: results from a randomised trial including 800+ recreational runners – 13/07/2021
Laurent Malisoux and 4 other authors -
Effect of shoe cushioning on landing impact forces and spatiotemporal parameters during running – 03/07/2021
Laurent Malisoux and 4 other authors -
Associations of physical activity level and variability with 6-month weight change among 26,935 users of connected devices – 15/04/2021
Guy Fagherazzi and 6 other authors -
Motion-control shoes reduce the risk of pronation-related pathologies in recreational runners – 01/03/2021
Tine Marieke Willems and 4 other authors -
Motivational Interviewing to Increase Physical Activity Behavior in Cancer Patients – 01/02/2021
Laurent Malisoux and 5 other authors -
Advanced analytical methods to assess physical activity behaviour using accelerometer raw time series data – 01/12/2020
Laurent Malisoux and 4 other authors -
Can the “Appropriate” footwear prevent injury in leisure-time running? evidence versus beliefs – 01/12/2020
Laurent Malisoux and 1 other author -
Running shoes and running injuries – 01/11/2020
Daniel Theisen and 2 other authors -
Physical ergonomics of distance running footwear – 03/09/2020
Laurent Malisoux and 1 other author -
Randomised controlled trials (RCTs) in sports injury research – 01/01/2020
Rasmus Oestergaard Nielsen and 7 other authors -
Motivational Interviewing to Increase Physical Activity Behavior in Cancer Patients – 01/01/2020
Laurent Malisoux and 7 other authors -
The stiffness response of type IIa fibres after eccentric exercise-induced muscle damage is dependent on ACTN3 r577X polymorphism – 21/04/2019
Siacia Broos and 6 other authors -
Challenges and benefits of integrating diverse sampling strategies in the observation of cardiovascular risk factors (ORISCAV-LUX 2) study – 04/02/2019
Alaa Al Kerwi and 17 other authors -
Mortality prediction of 35 frailty scores in a 7-years follow-up study in elderly general population – 01/11/2016
Gloria Aguayo and 8 other authors -
Effect Of Plyometric Training On Mechanical Properties Of Human Single Muscle Fibres – 03/06/2005
Laurent Malisoux and 2 other authors -
Ten-Year Trends in Healthcare Use and Costs for People Living with Diabetes in France – 25/03/2026
Guy Fagherazzi and 9 other authors -
Association of eating disorders and/or insulin omission with impaired glycaemic control in persons living with type 1 diabetes – 19/03/2026
Patrick Jean Ritz and 24 other authors -
Translating AI research into reality – 16/03/2026
Samantha Salvi Cruz and 49 other authors -
Voice Signatures of Momentary Psychological Stress in Real-Life Environments – 01/01/2026
Guy Fagherazzi and 6 other authors -
Interactions between genetic predisposition to obesity, insulin resistance and type 2 diabetes risk, and food or beverage intake for incident type 2 diabetes – 01/03/2026
Nicholas J. Wareham and 31 other authors -
Ethical Challenges in Automated Insulin Delivery and Emerging Diabetes Technologies – 01/02/2026
Ralph El Khoury and 2 other authors -
Psychosocial burden in type 1 diabetes – 29/01/2026
Gloria Aguayo and 10 other authors -
Data-driven clinical decision support tool for diagnosing mild cognitive impairment in Parkinson’s disease – 12/01/2026
Stefano Sapienza and 55 other authors -
Association between transportation noise exposure and type 2 diabetes risk in a French prospective cohort – 02/12/2025
Elodie Faure and 16 other authors -
Severity of complications is associated with impaired health-related quality of life in people with type 1 diabetes – 01/01/2025
Sara Barraud and 20 other authors -
Beyond overweight, visceral adiposity is associated with estimation of cardiovascular risk in patients living with type 1 diabetes – 01/12/2025
Laurence Salle and 17 other authors -
Time below range alone is insufficient to identify severe hypoglycaemia risk in type 1 diabetes—the critical role of hypoglycaemia awareness – 09/09/2025
Dulce Canha and 15 other authors -
A nationwide 12-month observatory of automated insulin delivery shows improved glucose control, sustained adoption, and reduced acute severe events – 01/01/2025
J. P. Riveline and 49 other authors -
Author Correction – 07/11/2025
Stefano Sapienza and 55 other authors -
Co-occurrence of memory impairment and fatigue distinguishes post COVID from pandemic-related health effects in the 4-year CON-VINCE cohort study – 27/10/2025
Dmitry Bulaev and 85 other authors -
Nationwide Trends in Type 1 and Type 2 Diabetes in France (2010–2019) – 01/01/2025
M. Joubert and 9 other authors -
Geographic environments, daily activities and stress in Luxembourg (the FragMent study) – 02/09/2025
Camille Perchoux and 14 other authors -
Role of the exposome in mental disorders – 18/08/2025
Claudia Gutierrez Ortiz and 4 other authors -
Immune cell–adipose tissue crosstalk in metabolic diseases with a focus on type 1 diabetes – 04/06/2025
Rasheed Ahmad and 6 other authors -
Interpretable Machine Learning for Cross-Cohort Prediction of Motor Fluctuations in Parkinson’s Disease – 22/04/2025
Rejko Krüger and 54 other authors -
Voice Assessment and Vocal Biomarkers in Heart Failure – 24/04/2025
Fabian Kerwagen and 10 other authors -
Multi-cohort machine learning identifies predictors of cognitive impairment in Parkinson’s disease – 26/07/2025
Lukas Pavelka and 55 other authors -
A Functionally-Grounded Benchmark Framework for XAI Methods – 14/07/2025
Dulce Canha and 3 other authors -
Musculoskeletal disorders in type 1 diabetes – 23/04/2025
Noémie Topalian and 19 other authors -
Racial disparities in continuous glucose monitoring-based 60-min glucose predictions among people with type 1 diabetes – 30/06/2025
Adam Hulman and 8 other authors -
Daily mobility, activity and environmental determinants of stress in ecological momentary assessment (EMA) and GPS studies – 27/06/2025
Noémie Topalian and 2 other authors -
Master protocols in vocal biomarker development to reduce variability and advance clinical precision – 27/06/2025
Yaël Bensoussan and 4 other authors -
Penetrance of Parkinson’s disease in GBA1 carriers depends on variant severity and polygenic background – 12/06/2025
Zied Landoulsi and 54 other authors -
Sex-specific progression of Parkinson’s disease – 01/06/2025
Anne-Marie Hanff and 9 other authors -
Trajectories of persisting Covid- 19 symptoms up to 24 months after acute infection – 25/04/2025
Aurélie Fischer and 8 other authors -
Cross-sectional analysis of eating disorder risk and risk correlates in candidates for bariatric surgery from the BariPredict cohort – 01/04/2025
Fawaz Alzaid and 11 other authors -
The effect of automated insulin delivery system use on diabetes distress in people with type 1 diabetes and their caregivers – 26/12/2024
Gloria Aguayo and 7 other authors -
Interactive Panel Summaries of the 2024 Voice AI Symposium – 27/03/2025
Jean Christophe Bélisle-Pipon and 32 other authors -
Association of LEF1-AS1 with cardiovascular and neurological complications of COVID-19 – 22/12/2024
Yvan Devaux and 8 other authors -
Clinical phenotyping of people living with type 1 diabetes according to their levels of diabetes-related distress – 12/08/2024
Guy Fagherazzi and 15 other authors -
Readiness of adults with type 1 diabetes and diabetes caregivers for diabetes distress monitoring using a voice-based digital health solution – 02/01/2025
Gloria Aguayo and 8 other authors -
A voice-based algorithm can predict type 2 diabetes status in USA adults – 19/12/2024
Guy Fagherazzi and 8 other authors -
Exploring Vocal Biomarkers For Disease Screening and Health Monitoring – 18/12/2024
Abir Elbeji -
Fear of hypoglycemia and sleep in children with type 1 diabetes and their parents – 16/12/2024
Ulrike Schierloh and 9 other authors -
Development of a digital health solution based on vocal biomarkers to remotely monitor frequent symptoms in real-life in people with Covid-19 or Long Covid. – 11/12/2024
Aurélie Fischer -
The Imperative of Voice Data Collection in Clinical Trials – 13/11/2024
Guy Fagherazzi and 1 other author -
Worldwide trends in diabetes prevalence and treatment from 1990 to 2022 – 13/11/2024
Bin Zhou and 49 other authors -
Digital voice-based biomarker for monitoring respiratory quality of life – 01/10/2024
Vladimir Despotovic and 6 other authors -
Dopamine Pathway and Parkinson’s Risk Variants Are Associated with Levodopa-Induced Dyskinesia – 01/01/2024
Ziv Gan-Or and 50 other authors -
Prediction of COVID-19 severity using machine learning – 01/10/2024
Yvan Devaux and 29 other authors -
Sex inequalities in cardiovascular risk factors and their management in primary prevention in adults living with type 1 diabetes in Germany and France – 16/09/2024
Emmanuel Cosson and 9 other authors -
Co-design of a voice-based app to monitor long COVID symptoms with its end-users – 09/09/2024
Aurélie Fischer and 8 other authors -
Integrating digital gait data with metabolomics and clinical data to predict outcomes in Parkinson’s disease – 06/09/2024
Enrico Glaab and 55 other authors -
Can we screen for type 2 diabetes using voice? – 01/09/2024
Guy Fagherazzi and 8 other authors -
Levodopa-induced dyskinesia in Parkinson’s disease – 01/09/2024
Rebecca Ting Jiin Loo and 54 other authors -
Multimodal Fusion for Vocal Biomarkers Using Vector Cross-Attention – 01/09/2024
Vladimir Despotovic and 3 other authors -
The transformative role of artificial intelligence in diabetes care and research – 27/07/2024
Guy Fagherazzi and 4 other authors -
Digital Vocal Biomarker of Smoking Status Using Ecological Audio Recordings – 03/07/2024
Guy Fagherazzi and 3 other authors -
Mixed effects models but not t-tests or linear regression detect progression of apathy in Parkinson’s disease over seven years in a cohort – 24/08/2024
Anne-Marie Hanff and 54 other authors -
Heterogeneity of glycaemic phenotypes in type 1 diabetes – 23/05/2024
Guy Fagherazzi and 29 other authors -
The impact of patient registration on utilisation and quality of care – 12/07/2024
Valerie Moran and 6 other authors -
Cardiovascular history and risk of idiopathic Parkinson’s disease – 08/07/2024
Shubhra Acharya and 47 other authors -
Smartphone use and cerebro-cardio-vascular health – 01/07/2024
Yvan Devaux and 2 other authors -
Assessment of the Impact of Subcutaneous Catheter Change on Glucose Control in Patients with Type 1 Diabetes Treated by Insulin Pump in Open- and Closed-Loop Modes – 01/01/2024
J. P. Riveline and 9 other authors -
A Voice-Based AI Algorithm Can Predict Type 2 Diabetes Status-Findings from the Colive Voice Study on US Adult Participants – 14/06/2024
Abir Elbeji and 8 other authors -
Converging peripheral blood microRNA profiles in Parkinson’s disease and progressive supranuclear palsy – 31/05/2024
Lukas Pavelka and 126 other authors -
Development of a long noncoding RNA-based machine learning model to predict COVID-19 in-hospital mortality – 20/05/2024
Yvan Devaux and 34 other authors -
Automatic detection of sleepiness-related symptoms and syndromes using voice and speech biomarkers – 01/05/2024
Vincent P Martin and 2 other authors -
The use of an automated insulin delivery system is associated with a reduction in diabetes distress and improvement in quality of life in people with type 1 diabetes – 01/01/2024
Yves Reznik and 7 other authors -
Why Voice Biomarkers of Psychiatric Disorders are not used in Clinical Practice? Deconstructing the Myth of the Need for Objective Diagnoses – 01/05/2024
Jean Luc Rouas and 1 other author -
Gut microbiome is not associated with mild cognitive impairment in Parkinson’s disease – 06/04/2024
Velma T.E. Aho and 55 other authors -
Objective evaluation of excessive daytime sleepiness – 23/02/2024
Jacques Taillard and 4 other authors -
Sleepiness should be reinvestigated through the lens of clinical neurophysiology – 23/02/2024
Jean Arthur Micoulaud-Franchi and 5 other authors -
Comprehensive blood metabolomics profiling of Parkinson’s disease reveals coordinated alterations in xanthine metabolism – 19/03/2024
Elisa Gómez De Lope and 133 other authors -
Automatic detection of sleepiness-related symptoms and syndromes using voice and speech biomarkers – 18/03/2024
Vincent P Martin and 2 other authors -
Creation of a pandemic memory by tracing COVID-19 infections and immunity in Luxembourg (CON-VINCE) – 09/02/2024
Olena Tsurkalenko and 30 other authors -
ALTRUIST – 05/02/2024
Guy Fagherazzi and 3 other authors -
Education as Risk Factor of Mild Cognitive Impairment – 01/01/2024
Matthias Klee and 137 other authors -
Development and validation of a Type 1 and Type 2 diabetes-specific patient-reported experience measure e-questionnaire – 01/01/2024
Coline Hehn and 11 other authors -
Relationship between multimorbidity, functional limitation, and quality of life among middle-aged and older adults – 01/01/2023
Piotr Wilk and 10 other authors -
Luxembourg Parkinson’s study -comprehensive baseline analysis of Parkinson’s disease and atypical parkinsonism – 19/12/2023
Rejko Krüger and 25 other authors -
Author Correction – 18/12/2023
Sinthuja Pachchek and 46 other authors -
Use of artificial intelligence methods for the analysis of real-world and social media data in digital epidemiology – 05/12/2023
Charline Bour -
The role of multimorbidity and socio-economic characteristics as potential risk factors for Long Covid – 01/12/2023
Piotr Wilk and 6 other authors -
Novel, computational IgE-clustering in a population-based cross-sectional study: Mapping the allergy burden – 01/12/2023
Rebecca Czolk and 10 other authors -
Accurate long-read sequencing identified GBA1 as major risk factor in the Luxembourgish Parkinson’s study – 23/11/2023
Sinthuja Pachchek and 131 other authors -
Sex-specific progression of Parkinson’s disease: A longitudinal mixed-models analysis – 01/11/2023
Anne-Marie Hanff and 9 other authors -
Association between endometriosis and risk of type 2 diabetes – 28/07/2023
Marina Kvaskoff and 5 other authors -
Mixed-methods study protocol to identify expectations of people with type 1 diabetes and their caregivers about voice-based digital health solutions to support the management of diabetes distress – 13/09/2023
Gloria Aguayo and 5 other authors -
Catheter changes lead to increased hyperglycaemia in both open and closed loop modes: real-life evidence from pump users with type 1 diabetes – 04/09/2023
Jean-Baptiste Julla and 10 other authors -
Device-measured physical activity and sedentary time in a national sample of Luxembourg residents: moving beyond the traditional metrics in the ORISCAV-LUX 2 study – 01/09/2023
Laurent Malisoux and 3 other authors -
Substituting device-measured sedentary time with alternative 24-hour movement behaviours: compositional associations with adiposity, cardiometabolic risk and arterial stiffness in the ORISCAV-LUX 2 study – 01/09/2023
Paul J Collings and 4 other authors -
Novel, computational IgE-clustering in a population-based cross-sectional study – 27/07/2023
Annette Kuehn and 10 other authors -
O.3.2-2 Substituting device-measured sedentary time with alternative 24-hour movement behaviours – 01/09/2023
Paul James Collings and 4 other authors -
The Long COVID experience from a patient’s perspective – 17/08/2023
Guy Fagherazzi and 4 other authors -
Association of diet quality indices with serum and metabolic biomarkers in participants of the ORISCAV-LUX-2 study – 01/01/2023
Farhad Vahid and 28 other authors -
Interaction between plasma phospholipid odd-chain fatty acids and GAD65 autoantibodies on the incidence of adult-onset diabetes – 10/06/2023
Anna Maria Lampousi and 22 other authors -
Technologies will not make diabetes disappear – 01/07/2023
Guy Fagherazzi -
Validation of a Parkinson’s disease questionnaire-39-based functional mobility composite score (FMCS) in people with Parkinson’s disease – 17/05/2023
Anne-Marie Hanff and 6 other authors -
Co-Design of a Voice-Based Digital Health Solution to Monitor Persisting Symptoms Related to COVID-19 (UpcomingVoice Study) – 19/06/2023
Aurélie Fischer and 6 other authors -
Profiles of Glycemic Control in People with Type 1 Diabetes Using CGM—A Clustering Approach in the SFDT1 Study – 01/06/2023
Emmanuel Cosson and 6 other authors -
Prevalence and description of skin reactions associated with adhesives in diabetes technology devices in an adult population – 24/05/2023
J. P. Riveline and 14 other authors -
A nutritional biomarker score of the Mediterranean diet and incident type 2 diabetes – 27/04/2023
Nita G. Forouhi and 33 other authors -
Patients’ and caregivers’ perceptions of bariatric surgery – 18/04/2023
Sébastien Czernichow and 10 other authors -
Substituting device-measured sedentary time with alternative 24-hour movement behaviours – 04/04/2023
Laurent Malisoux and 4 other authors -
Prevalence and Description of the Skin Reactions Associated with Adhesives in Diabetes Technology Devices in an Adult Population – 24/02/2023
J. P. Riveline and 14 other authors -
Association between use of psychotropic medications prior to SARS-COV-2 infection and trajectories of COVID-19 recovery – 16/03/2023
Gloria Aguayo and 5 other authors -
Altered infective competence of the human gut microbiome in COVID-19 – 09/03/2023
Laura de Nies and 28 other authors -
Characterizing the adult exposome in men and women from the general population – 26/01/2023
Maria Ruiz and 8 other authors -
Diabetes registries and high-quality diabetes care – 01/02/2023
Kamlesh Khunti and 18 other authors -
Correction – 31/01/2023
Gloria Aguayo and 10 other authors -
Machine learning for predicting neurodegenerative diseases in the general older population – 11/01/2023
Gloria Aguayo and 10 other authors -
PRODIAB – 01/01/2023
Nicolas Naïditch and 6 other authors -
Les réseaux sociaux au service de la recherche sur le diabète – 01/01/2023
Charline Bour and 1 other author -
Towards a European “Cohort Moonshot” – 01/01/2023
Guy Fagherazzi -
Associations Between Wearable-Specific Indicators of Physical Activity Behaviour and Insulin Sensitivity and Glycated Haemoglobin in the General Population – 12/12/2022
Anne Backes and 29 other authors -
Device-measured physical activity and sedentary time in a national sample of Luxembourg residents – 01/12/2022
Laurent Malisoux and 3 other authors -
Long COVID Classification – 30/11/2022
Aurélie Fischer and 8 other authors -
Identifying Profiles and Symptoms of Patients With Long COVID in France – 22/11/2022
Amélia Déguilhem and 10 other authors -
Vocal biomarker predicts fatigue in people with COVID-19 – 22/11/2022
Guy Fagherazzi and 7 other authors -
Association of miR-144 levels in the peripheral blood with COVID-19 severity and mortality – 21/11/2022
Fabio Martelli and 17 other authors -
Recommendations for Successful Implementation of the Use of Vocal Biomarkers for Remote Monitoring of COVID-19 and Long COVID in Clinical Practice and Research – 15/11/2022
Aurélie Fischer and 3 other authors -
Discovery and Analytical Validation of a Vocal Biomarker to Monitor Anosmia and Ageusia in Patients with COVID-19 – 07/09/2022
Guy Fagherazzi and 6 other authors -
A voice-based biomarker for monitoring symptom resolution in adults with COVID-19 – 20/10/2022
Guy Fagherazzi and 8 other authors -
Global diabetes burden – 01/10/2022
Guy Fagherazzi and 5 other authors -
Association between functional limitation and quality of life among older adults with multimorbidity in Luxembourg – 01/10/2022
Piotr Wilk and 9 other authors -
How multimorbidity and socio-economic factors affect Long COVID: Evidence from European Countries – 01/10/2022
Piotr Wilk and 6 other authors -
Analysing breast cancer survivors’ acceptance profiles for using an electronic pillbox connected to a smartphone application using Seintinelles, a French community-based research tool – 27/09/2022
Catherine Goetzinger and 6 other authors -
Publisher Correction – 02/09/2022
Lukas Pavelka and 47 other authors -
Association between endometriosis and risk of type 2 diabetes: results of the E3N prospective cohort study – 01/09/2022
P. Vaduva and 4 other authors -
Precision Health – 01/09/2022
Guy Fagherazzi and 11 other authors -
Precision Health – 01/09/2022
Guy Fagherazzi and 11 other authors -
Precision Health – 01/09/2022
Guy Fagherazzi and 11 other authors -
Age at onset as stratifier in idiopathic Parkinson’s disease – effect of ageing and polygenic risk score on clinical phenotypes – 09/08/2022
Lukas Pavelka and 50 other authors -
Long COVID Symptomatology After 12 Months and Its Impact on Quality of Life According to Initial Coronavirus Disease 2019 Disease Severity – 05/08/2022
Guy Fagherazzi and 8 other authors -
Design and validation of a patient-reported Functional Mobility Composite Score (FMCS) in people with Parkinson’s disease – 20/07/2022
Anne-Marie Hanff and 6 other authors -
Extraction of Explicit and Implicit Cause-Effect Relationships in Patient-Reported Diabetes-Related Tweets From 2017 to 2021 – 19/07/2022
Adrian Ahne and 8 other authors -
Emulating a virtual digital cohort study based on social media data as a complementary approach to traditional epidemiology – 24/05/2022
Guy Fagherazzi and 2 other authors -
The Nutri-Score nutrition label – 27/07/2021
Serge Hercberg and 6 other authors -
Intermittent Scanning Glucose Monitoring or Predicted Low Suspend Pump Treatment – 31/05/2022
Ulrike Schierloh and 8 other authors -
Association of neighbourhood disadvantage and individual socioeconomic position with all-cause mortality – 01/05/2022
Ana Isabel Ribeiro and 49 other authors -
Associations between physical activity prior to infection and COVID-19 disease severity and symptoms – 29/04/2022
Laurent Malisoux and 5 other authors -
Impact of intra-category food substitutions on the risk of type 2 diabetes – 01/01/2021
Moufidath Adjibade and 7 other authors -
Identification of vocal biomarkers for screening diabetes and monitoring health of people with diabetes: preliminary results from the COLIVE voice study – 25/04/2022
Abir Elbeji and 3 other authors -
Combinatorial analysis reveals highly coordinated early-stage immune reactions that predict later antiviral immunity in mild COVID-19 patients – 01/01/2022
Christophe Capelle and 23 other authors -
Interim analysis of COVID-19 vaccine effectiveness against Severe Acute Respiratory Infection due to laboratory-confirmed SARS-CoV-2 among individuals aged 30 years and older, ECDC multi-country study – second update – 14/03/2022
Nassera Aouali and 4 other authors -
Attitudes and Expectations of Clinical Research Participants Toward Digital Health and Mobile Dietary Assessment Tools – 09/03/2022
Florent Schäfer and 6 other authors -
Associations Between Device-Measured Physical Activity and Glycemic Control and Variability Indices Under Free-Living Conditions – 14/12/2021
Douae El Fatouhi and 7 other authors -
Comparison of Deep Neural Networks and Regularised Cox Regression Models in the Prediction of Neurodegenerative Diseases in the General Older Population – 01/03/2022
Gloria Aguayo and 10 other authors -
Association between Dietary Factors and Constipation in Adults Living in Luxembourg and Taking Part in the ORISCAV-LUX 2 Survey – 28/12/2021
Maurane Rollet and 17 other authors -
Improving Diabetes-Related Biomedical Literature Exploration in the Clinical Decision-making Process via Interactive Classification and Topic Discovery – 01/01/2022
Adrian Ahne and 4 other authors -
Objective and subjective sleep measures are associated with HbA1c and insulin sensitivity in the general population – 20/05/2021
Gloria Aguayo and 10 other authors -
Cardiometabolic Health Associated with Exposures to Mixtures of Chemical Pollutants: Results from the EHES-LUX Study – 01/01/2022
Giovana M. Ciprián and 6 other authors -
Long COVID Classification: Findings from a Clustering Analysis in the Predi-COVID Cohort Study – 01/01/2022
Aurélie Fischer and 8 other authors -
Estimated visceral adiposity is associated with risk of cardiometabolic conditions in a population based study – 01/12/2021
Maria Ruiz and 5 other authors -
Methods to Generate Innovative Research Ideas and Improve Patient and Public Involvement in Modern Epidemiological Research – 01/12/2021
Gloria Aguayo and 9 other authors -
Allergy burden in luxembourg: A population-based, cross-sectional health survey paralleled by component-resolved-IgE-diagnosis – 15/11/2021
Rebecca Czolk and 13 other authors -
Identifying causal associations in tweets using deep learning: Use case on diabetes-related tweets from 2017-2021 – 01/11/2021
Adrian Ahne and 8 other authors -
Intérêt d’une approche géostatistique pour analyser les disparités géographiques de la couverture vaccinale contre les papillomavirus humains en France – 30/10/2021
L. Ribassin-Majed and 6 other authors -
Detection of COVID-19 from voice, cough and breathing patterns – 01/11/2021
Vladimir Despotovic and 4 other authors -
Unraveling the concepts of distress, burnout, and depression in type 1 diabetes – 01/10/2021
Gloria Aguayo and 7 other authors -
Systems and Longitudinal Analysis Reveals Early-Stage and Highly Coordinated Immune Responses in Mild COVID-19 Patients – 30/09/2021
Christophe Capelle and 22 other authors -
Deep immune profiling reveals early-stage and highly coordinated immune responses in mild COVID-19 patients – 02/09/2021
Feng Q Hefeng and 22 other authors -
Digital Health Interventions among People Living with Frailty – 01/01/2021
Gloria Aguayo and 5 other authors -
Musculoskeletal disorders in type 1 diabetes – 01/09/2021
S. Picard and 2 other authors -
Author Correction – 25/08/2021
Maria Ruiz and 5 other authors -
Towards precision cardiometabolic prevention – 06/08/2021
Guy Fagherazzi and 10 other authors -
Rapport du groupe de travail en charge de la réalisation d’une étude indépendante au sujet des clusters observés dans certaines structures d’hébergement pour personnes âgées – 10/07/2021
Tamir Abdelrahman and 7 other authors -
Association of Migraine With Incident Hypertension After Menopause – 06/07/2021
Conor James Macdonald and 6 other authors -
Is the Consensual Threshold for Defining High Glucose Variability Implementable in Clinical Practice? – 07/06/2021
Jean Baptiste Julla and 12 other authors -
Type 2 diabetes and its characteristics are associated with poor oral health – 23/06/2021
Guy Fagherazzi and 6 other authors -
Voice for Health – 01/06/2021
Guy Fagherazzi and 3 other authors -
Deep digital phenotyping in type 1 diabetes – 01/06/2021
Guy Fagherazzi -
The use of social media for health research purposes – 27/05/2021
Guy Fagherazzi and 4 other authors -
SARS-CoV-2 transmission risk from asymptomatic carriers – 01/05/2021
Paul Wilmes and 14 other authors -
The long-run effects of war on health – 01/05/2021
Julia Mink and 2 other authors -
Associations of physical activity level and variability with 6-month weight change among 26,935 users of connected devices – 15/04/2021
Guy Fagherazzi and 6 other authors -
Systematic review and network meta-analysis of the efficacy of existing treatments for patients with recurrent glioblastoma – 09/04/2021
Anna Schritz and 6 other authors -
Gonadal hormonal factors before menopause and incident type 2 diabetes in women: A 22-year follow-up of 83 799 women from the E3N cohort study – 01/04/2021
Guy Fagherazzi and 7 other authors -
Scoping review protocol on the use of social media for health research purposes – 11/02/2021
Guy Fagherazzi and 4 other authors -
Beliefs and attitudes towards an ehealth tool to improve medication adherence in breast cancer survivors – 01/02/2021
Catherine Goetzinger and 6 other authors -
Challenges and perspectives for the future of diabetes epidemiology in the era of digital health and artificial intelligence – 01/01/2021
Guy Fagherazzi -
Micronutrients and markers of oxidative stress and inflammation related to cardiometabolic health – 22/12/2020
Maria Ruiz and 8 other authors -
Plasma Vitamin C and type 2 diabetes – 01/01/2021
Nicholas J. Wareham and 41 other authors -
Advanced analytical methods to assess physical activity behaviour using accelerometer raw time series data – 01/12/2020
Laurent Malisoux and 4 other authors -
Associations between early-life food deprivation during World War II and risk of hypertension and type 2 diabetes at adulthood – 01/12/2020
Guy Fagherazzi and 4 other authors -
Associations between physical activity and incident hypertension across strata of body mass index – 01/12/2020
Marie Christine Boutron-Ruault and 7 other authors -
Beyond the map – 01/12/2020
Yohan Fayet and 7 other authors -
Genome-wide association analysis of type 2 diabetes in the EPIC-InterAct study – 01/12/2020
Nicholas J. Wareham and 32 other authors -
Intricate relationships between frailty and diabetes – 01/12/2020
Gloria Aguayo and 1 other author -
Protocol for a prospective, longitudinal cohort of people with COVID-19 and their household members to study factors associated with disease severity – 23/11/2020
Guy Fagherazzi and 13 other authors -
Mapping and Modeling of Discussions Related to Gastrointestinal Discomfort in French-Speaking Online Forums – 01/11/2020
Florent Schäfer and 8 other authors -
Replacement of red and processed meat with other food sources of protein and the risk of type 2 diabetes in European populations – 01/11/2020
Daniel B. Ibsen and 42 other authors -
The potential benefits of the FreeStyle® Libre™ 2 alarm system on the mental load of people with diabetes – 01/11/2020
Caroline Guillot and 1 other author -
The association between circulating 25-hydroxyvitamin D metabolites and type 2 diabetes in European populations – 16/10/2020
Ju Sheng Zheng and 49 other authors -
Statins and breast cancer risk in a cohort of postmenopausal women – 01/10/2020
Laure Dossus and 8 other authors -
Combination disease prevention in prisons: a comprehensive programme in Luxembourg – 27/07/2020
Patrick Hoffmann and 10 other authors -
Digital health strategies to fight COVID-19 worldwide – 01/06/2020
Guy Fagherazzi and 4 other authors -
Multiomics for Risk Stratification in Atherosclerotic Cardiovascular Disease – 25/03/2026
Liv Tybjærg Nordestgaard and 12 other authors -
Shared splicing dysregulation in heart failure associated with dilated and ischemic cardiomyopathy and spatial specificity across cardiac regions – 24/03/2026
Sandra Martins and 10 other authors -
Pros and cons of Swedish law on intellectual property rights – 13/03/2026
Ljubica Matic and 2 other authors -
RNA modifications in peripheral blood are associated with acute coronary syndrome – 04/03/2026
Yvan Devaux and 10 other authors -
Blood N1-methyladenosine (m1A) RNA modification and outcome after cardiac arrest – 25/02/2026
Yvan Devaux and 6 other authors -
Relevance of pre-analytical factors in multiomics – 17/01/2026
Marie Mardal and 20 other authors -
Do nurses and allied professionals need to upskill to meet developments in RNA therapeutics for CVD? – 09/01/2026
Faye Forsyth and 1 other author -
Direct RNA sequencing identified solute carrier family 2 member 1 to improve neurological outcome prediction after cardiac arrest – 07/01/2026
Yvan Devaux and 16 other authors -
prm-PASEF-Based Quantification and Isomeric Model for Extended Coverage of Human Plasma Lipidome in Parkinson’s Disease – 11/11/2025
Laura Bindila and 6 other authors -
Biological Sex and Cardiovascular Disease Prevention in Systemic Arterial Hypertension – 11/09/2025
Eva Gerdts and 6 other authors -
Harnessing multiomics technologies and machine learning for advancing personalized theranostic approaches in atherosclerosis – 29/09/2025
Soumaya Ben-Aicha and 5 other authors -
Co-occurrence of memory impairment and fatigue distinguishes post COVID from pandemic-related health effects in the 4-year CON-VINCE cohort study – 27/10/2025
Dmitry Bulaev and 85 other authors -
Bottlenecks in advancing and applying multiomic data integration-common data resources as rate-limiting drivers-the high-impact use case of atherosclerotic cardiovascular disease – 10/10/2025
Stephanie Bezzina Wettinger and 22 other authors -
Established and Emerging Roles of Epigenetic Regulation in Diabetic Cardiomyopathy – 01/09/2025
Adam Russell-Hallinan and 11 other authors -
Multiomics in atherosclerotic cardiovascular disease – 26/06/2025
Liv Tybjærg Nordestgaard and 12 other authors -
Prognostic and predictive microRNA panels for heart failure patients with reduced or preserved ejection fraction – 07/07/2025
Reza Parvan and 8 other authors -
A protocol for microRNA extraction from gastrointestinal digesta – 01/06/2025
Torsten Bohn and 2 other authors -
Deciphering immune dynamics in atherosclerosis – 01/01/2025
Rui Vitorino and 6 other authors -
Editorial – 01/01/2025
Miron Sopic and 4 other authors -
Epitranscriptomics in atherosclerosis – 01/06/2025
Victoria Stopa and 6 other authors -
Association of LEF1-AS1 with cardiovascular and neurological complications of COVID-19 – 22/12/2024
Yvan Devaux and 8 other authors -
Circulating levels of miR-20b-5p are associated with survival in cardiogenic shock – 17/01/2025
Tuomas Mäntylä and 10 other authors -
Blood CD45
+/CD3
+ lymphocyte-released extracellular vesicles and mortality in hospitalized patients with coronavirus disease 2019. – 01/01/2024
Lina Badimon and 12 other authors -
IMMUnity Unveiled – 23/01/2025
Inês Figueira and 14 other authors -
Biogenesis and mechanisms of action of ncRNAs – 01/01/2025
Anne Yaël Nossent and 6 other authors -
Essentials of transcriptomic methods – 01/01/2025
Victoria Stopa and 8 other authors -
Transcriptomics in Atherosclerosis – 01/01/2025
Yvan Devaux and 1 other author -
Circular RNA regulatory role in pathological cardiac remodelling – 01/01/2024
Fabio Martelli and 14 other authors -
EDITORIAL for BJP themed issue “noncoding RNA therapeutics” – 21/11/2024
Yvan Devaux and 2 other authors -
Targeting noncoding RNAs to treat atherosclerosis – 08/05/2024
Yvan Devaux and 7 other authors -
Navigating the path of reproducibility in microRNA-based biomarker research with ring trials – 01/01/2024
David de Gonzalo-Calvo and 2 other authors -
Increased [18F]FDG uptake in the infarcted myocardial area displayed by combined PET/CMR correlates with snRNA-seq-detected inflammatory cell invasion – 26/06/2024
Dominika Lukovic and 19 other authors -
Prediction of COVID-19 severity using machine learning – 01/10/2024
Yvan Devaux and 29 other authors -
Multiomic biomarkers after cardiac arrest – 27/09/2024
Yvan Devaux and 9 other authors -
Guidelines for mitochondrial RNA analysis – 10/09/2024
Yvan Devaux and 11 other authors -
Machine learning for catalysing the integration of noncoding RNA in research and clinical practice – 18/07/2024
George Kararigas and 7 other authors -
miR-619-5p and cardiogenic shock in patients with ST-segment elevation myocardial infarction – 01/01/2024
Lina Badimon and 8 other authors -
Non-coding RNAs as therapeutic targets and biomarkers in ischaemic heart disease – 01/01/2024
Costanza Emanueli and 7 other authors -
Biomarkers of food intake and their relevance to metabolic syndrome – 01/01/2024
Torsten Bohn and 3 other authors -
Cardiovascular history and risk of idiopathic Parkinson’s disease – 08/07/2024
Shubhra Acharya and 47 other authors -
Smartphone use and cerebro-cardio-vascular health – 01/07/2024
Yvan Devaux and 2 other authors -
CD66b+/CD68+ circulating extracellular vesicles, lactate dehydrogenase and neutrophil-to-lymphocyte ratio can differentiate coronavirus disease 2019 severity during and after infection – 01/07/2024
Lina Badimon and 6 other authors -
Addressing the unsolved challenges in microRNA-based biomarker development – 28/04/2024
David de Gonzalo-Calvo and 15 other authors -
Development of a long noncoding RNA-based machine learning model to predict COVID-19 in-hospital mortality – 20/05/2024
Yvan Devaux and 34 other authors -
The challenges of research data management in cardiovascular science – 01/01/2023
Peter Kohl and 24 other authors -
Leveraging epitranscriptomics for cardiovascular disease theranostics – 08/01/2024
Yvan Devaux and 2 other authors -
Exploring pulmoprotection in COVID-19 – 12/03/2024
Miron Sopic -
Recommendations for detection, validation, and evaluation of RNA editing events in cardiovascular and neurological/neurodegenerative diseases – 12/03/2024
Anne Yaël Nossent and 12 other authors -
Multicomponent (bio)markers for obesity risk prediction – 08/03/2024
Torsten Bohn and 21 other authors -
Evolution of journal clubs – 01/03/2024
Yvan Devaux and 7 other authors -
The impact of government policies, funding, and networking to accelerate transatlantic cardiovascular research – 16/12/2023
Shubhra Acharya and 3 other authors -
Long noncoding RNAs in tissue repair – LncSSH1 – 15/12/2023
Shubhra Acharya -
Firalink – 01/12/2023
Yvan Devaux and 6 other authors -
Multiomics tools for improved atherosclerotic cardiovascular disease management – 01/01/2023
Miron Sopic and 8 other authors -
Transcriptomic research in atherosclerosis – 01/12/2023
Yvan Devaux and 10 other authors -
HCG18, LEF1AS1 and lncCEACAM21 as biomarkers of disease severity in the peripheral blood mononuclear cells of COVID-19 patients – 26/10/2023
Fabio Martelli and 18 other authors -
A Machine Learning Model Based on microRNAs for the Diagnosis of Essential Hypertension – 25/10/2023
Yvan Devaux and 5 other authors -
The science behind soft skills – 12/10/2023
Yvan Devaux and 17 other authors -
Association of diet quality indices with serum and metabolic biomarkers in participants of the ORISCAV-LUX-2 study – 01/01/2023
Farhad Vahid and 28 other authors -
HCG18, LEF1AS1 and lncCEACAM21 as biomarkers of disease severity in the Peripheral Blood Mononuclear Cells of COVID-19 patients – 07/07/2023
Fabio Martelli and 17 other authors -
Call for participation in the AtheroNET COST Action to implement multiomics in atherosclerotic cardiovascular disease research – 12/04/2023
Miron Sopic and 3 other authors -
Targeting regulatory RNAs at the heart of Parkinson’s disease – 08/03/2023
Yvan Devaux and 2 other authors -
RNA modification m6Am – 18/06/2023
Markéta Hlavackova and 4 other authors -
GATA3 as a Blood-Based RNA Biomarker for Idiopathic Parkinson’s Disease – 12/06/2023
Yvan Devaux and 4 other authors -
Cardiovascular complications of diabetes – 24/05/2023
Fabio Martelli and 14 other authors -
Integration of epigenetic regulatory mechanisms in heart failure – 04/05/2023
Yvan Devaux and 9 other authors -
The circadian clock remains intact, but with dampened hormonal output in heart failure – 17/04/2023
Linda W. Van Laake and 20 other authors -
Development of circulating microRNA-based biomarkers for medical decision-making – 02/11/2022
David de Gonzalo-Calvo and 5 other authors -
FIMICS – 01/01/2023
Yvan Devaux and 11 other authors -
Peripheral blood RNA biomarkers for cardiovascular disease from bench to bedside – 14/10/2021
Maarten Vanhaverbeke and 14 other authors -
Non-coding RNAs as prognostic biomarkers of cardiac arrest – 14/12/2022
Francesca Maria STEFANIZZI -
Diagnostic performance of microRNAs in the detection of heart failure with reduced or preserved ejection fraction – 01/01/2022
Gustavo José Justo da Silva and 5 other authors -
Association of miR-144 levels in the peripheral blood with COVID-19 severity and mortality – 21/11/2022
Fabio Martelli and 17 other authors -
Correction – 09/11/2022
Yvan Devaux and 12 other authors -
Circular RNAs to predict clinical outcome after cardiac arrest – 28/10/2022
Yvan Devaux and 12 other authors -
(Epi)transcriptomics in cardiovascular and neurological complications of COVID-19 – 28/07/2022
Yvan Devaux and 4 other authors -
Precision Health – 01/09/2022
Guy Fagherazzi and 11 other authors -
Precision Health – 01/09/2022
Guy Fagherazzi and 11 other authors -
Precision Health – 01/09/2022
Guy Fagherazzi and 11 other authors -
MicroRNAs as biomarkers in the brain-heart axis? – 11/07/2022
Yvan Devaux -
Methodological considerations for circulating long noncoding RNA quantification – 01/01/2022
David de Gonzalo-Calvo and 2 other authors -
Regulation of N6-Methyladenosine after Myocardial Infarction – 22/07/2022
Yvan Devaux and 10 other authors -
Noncoding RNAs in age-related cardiovascular diseases – 16/03/2022
Yvan Devaux and 11 other authors -
Dissecting the transcriptome in cardiovascular disease – 23/03/2021
Fabio Martelli and 14 other authors -
Consensus guidelines for the validation of qRT-PCR assays in clinical research by the CardioRNA consortium – 10/03/2022
Yvan Devaux and 10 other authors -
Editorial – 23/02/2022
David de Gonzalo-Calvo and 2 other authors -
Challenges of microRNA-based biomarkers in clinical application for cardiovascular diseases – 01/02/2022
David de Gonzalo-Calvo and 3 other authors -
Association between Dietary Factors and Constipation in Adults Living in Luxembourg and Taking Part in the ORISCAV-LUX 2 Survey – 28/12/2021
Maurane Rollet and 17 other authors -
Leveraging non-coding RNAs to fight cardiovascular disease – 21/12/2021
Emma Robinson and 3 other authors -
Relevance of N6-methyladenosine regulators for transcriptome – 01/11/2021
Costanza Emanueli and 4 other authors -
Non-coding RNAs and stem cells – 01/10/2021
Yvan Devaux and 2 other authors -
IMproving Preclinical Assessment of Cardioprotective Therapies (IMPACT) criteria – 13/09/2021
Sandrine Lecour and 49 other authors -
The circadian clock remains intact, but with dampened hormonal output in heart failure – 01/09/2021
Sandra Crnko and 14 other authors -
Cardiovascular RNA markers and artificial intelligence may improve COVID-19 outcome – 01/07/2021
Yvan Devaux and 18 other authors -
N6-Methyladenine in Eukaryotic DNA – 24/05/2021
Jonathan Turner and 12 other authors -
Conclusions and perspectives – 06/03/2021
Emma Louise Robinson and 1 other author -
Epigenetics in Cardiovascular Disease – 06/03/2021
Trygve Tollefsbol and 2 other authors -
Long noncoding RNAs and circular RNAs as heart failure biomarkers – 06/03/2021
Amela Jusic and 1 other author -
Preface – 06/03/2021
Yvan Devaux and 1 other author -
The Long Noncoding RNA Landscape of Cardiac Regeneration in Zebrafish – 01/03/2021
Yvan Devaux and 4 other authors -
Joining European Scientific Forces to Face Pandemics – 01/02/2021
Chiara Riganti and 21 other authors -
CDR132L – 07/01/2021
Yvan Devaux and 1 other author -
Increased mir-142 levels in plasma and atherosclerotic plaques from peripheral artery disease patients with post-surgery cardiovascular events – 02/12/2020
Loredan S. Niculescu and 6 other authors -
Noncoding RNAs implication in cardiovascular diseases in the COVID-19 era – 01/12/2020
Fabio Martelli and 5 other authors -
Regulation of microRNAs in high-fat diet induced hyperlipidemic hamsters – 01/12/2020
Loredan S. Niculescu and 6 other authors -
Association of mir-21-5p, mir-122-5p, and mir-320a-3p with 90-day mortality in cardiogenic shock – 01/11/2020
Mikko Hänninen and 11 other authors -
Circular RNAs as prognostic biomarkers after cardiac arrest – 01/11/2020
Francesca Maria STEFANIZZI and 4 other authors -
Non-coding rnas in the brain-heart axis – 02/09/2020
Yvan Devaux and 7 other authors -
Approaching sex differences in cardiovascular non-coding rna research – 02/07/2020
Susana Novella and 9 other authors -
Circulating levels of brain-enriched micrornas correlate with neuron specific enolase after cardiac arrest—a substudy of the target temperature management trial – 02/06/2020
Francesca Maria STEFANIZZI and 12 other authors -
Cardiomyocyte-Specific Cell-Free DNA as a Heart Failure Biomarker? – 01/06/2020
Yvan Devaux -
Call to action for the cardiovascular side of covid-19 – 14/05/2020
Yvan Devaux and 6 other authors -
RNAs in brain and heart diseases – 02/05/2020
Yvan Devaux and 4 other authors -
A year in the life of the EU-CardioRNA COST action – 01/05/2020
Emma Robinson and 49 other authors -
Mitochondrial noncoding RNA-regulatory network in cardiovascular disease – 01/05/2020
Yvan Devaux and 1 other author -
Epigenetics in non-classical monocytes support their pro-inflammatory gene expression – 01/05/2020
Loems Ziegler-Heitbrock and 8 other authors -
Copeptin as a marker of outcome after cardiac arrest – 28/04/2020
Joachim Düring and 15 other authors -
High-intensity aerobic training lowers blood pressure and modulates the renal renin-angiotensin system in spontaneously hypertensive rats – 02/04/2020
Octávio Luiz Franco and 8 other authors -
Potential clinical implications of mir-1 and mir-21 in heart disease and cardioprotection – 01/02/2020
Monika Bartekova and 3 other authors -
Regulatory RNAs in Heart Failure – 27/01/2020
Yvan Devaux and 9 other authors -
A role for m6A RNA methylation in heart failure development? – 01/01/2020
Yvan Devaux and 1 other author -
Transcriptomics research to improve cardiovascular healthcare – 01/01/2020
Yvan Devaux and 1 other author -
Les microARN sanguins pour prédire l’insuffisance cardiaque post-infarctus – 25/12/2019
Yvan Devaux -
Peripheral Blood RNA Levels of QSOX1 and PLBD1 Are New Independent Predictors of Left Ventricular Dysfunction after Acute Myocardial Infarction – 01/12/2019
Maarten Vanhaverbeke and 12 other authors -
Noncoding RNAs in Hypertension – 01/09/2019
Yvan Devaux and 1 other author -
MIR-574-5p – 02/08/2019
Yvan Devaux and 9 other authors -
Circulating microRNAs to predict heart failure after acute myocardial infarction in women – 01/08/2019
Yvan Devaux and 1 other author -
Immune cells as targets for cardioprotection – 01/06/2019
Ioanna Andreadou and 9 other authors -
Circulating levels of microRNA 423-5p are associated with 90 day mortality in cardiogenic shock – 01/02/2019
Toni Jäntti and 12 other authors -
Preface “MicroRNAs have it all” – 01/02/2019
Yvan Devaux -
The association between plasma miR-122-5p release pattern at admission and all-cause mortality or shock after out-of-hospital cardiac arrest – 02/01/2019
Patrik Gilje and 12 other authors -
Cyclin dependent kinase inhibitor 1 C is a female-specific marker of left ventricular function after acute myocardial infarction – 01/01/2019
Yvan Devaux and 7 other authors -
Catalyzing transcriptomics research in cardiovascular disease – 01/01/2019
Yvan Devaux and 49 other authors -
Circulating levels of miR-574-5p are associated with neurological outcome after cardiac arrest in women – 01/01/2019
Yvan Devaux and 11 other authors -
Regulation of microRNAs in coronary atherosclerotic plaque – 01/01/2019
Yvan Devaux and 9 other authors -
Diagnostic, prognostic and therapeutic uses of long noncoding RNAs for pathologies and toxicities inducing heart disorders – 20/12/2018
Huseyin Firat and 7 other authors -
Non-coding RNAs to aid in neurological prognosis after cardiac arrest – 18/12/2018
Yvan Devaux and 6 other authors -
Novel circular RNA biomarkers for heart failure – 06/12/2018
Yvan Devaux and 1 other author -
Biomarqueurs transcriptomiques sanguins des maladies cardiovasculaires – 30/11/2018
Adeline Boileau -
Biomarkers of left ventricular function after acute myocardial infarction: sex-biased differences – 16/11/2018
Torkia Lalem -
Noncoding RNAs in acute kidney injury – 01/11/2018
Timo Brandenburger and 3 other authors -
Long Noncoding RNAs and Cardiac Disease – 20/09/2018
Fabio Martelli and 3 other authors -
Practical guidelines for rigor and reproducibility in preclinical and clinical studies on cardioprotection – 01/09/2018
Hans Erik Bøtker and 29 other authors -
Cyclin dependent kinase inhibitor 1C (CDKN1C), a novel female-specific marker of left ventricular dysfunction after acute myocardial infarction – 01/08/2018
Torkia Lalem and 6 other authors -
Restoration of cardiac function after anaemia-induced heart failure in zebrafish – 01/08/2018
Isabelle Ernens and 2 other authors -
What’s new in prognostication after cardiac arrest – 01/06/2018
Yvan Devaux and 1 other author -
Non-coding RNAs and exercise – 01/05/2018
Yvan Devaux and 6 other authors -
Cardiac enriched lncRNAs as a novel class of biomarkers for heart condition and prediction of heart failure – 01/05/2018
Eric Schordan and 8 other authors -
Circular RNA MICRA predicts heart failure after acute myocardial infarction – 01/05/2018
Mélanie Vausort and 3 other authors -
Cyclin dependent kinase inhibitor 1 C is a female-specific marker of left ventricular function after acute myocardial infarction – 01/05/2018
Torkia Lalem and 7 other authors -
Endogenous heparin interferes with quantification of MicroRNAs by RT-qPCR – 01/05/2018
Yvan Devaux and 3 other authors -
Identification of circulating microRNAs from HTG EdgeSeq sequencing to predict outcome after acute myocardial infarction – 01/05/2018
Clarissa Pedrosa da Costa Gomes and 5 other authors -
Kruppel-like factors regulation in a zebrafish heart failure model – 01/05/2018
Antonio Salgado Somoza and 4 other authors -
Late heartbeat-evoked potentials are associated with survival after cardiac arrest – 01/05/2018
André Schulz and 6 other authors -
Times are changing: differential regulation of clock genes in mice and zebrafish with heart failure – 01/05/2018
Sandra Crnko and 7 other authors -
Realizing the therapeutic potential of novel cardioprotective therapies: The EU-CARDIOPROTECTION COST Action – CA16225 – 01/04/2018
Ioannou Andreadou and 49 other authors -
A 3-gene panel improves the prediction of left ventricular dysfunction after acute myocardial infarction – 01/03/2018
Yvan Devaux and 4 other authors -
Circular RNAs in the cardiovascular system – 01/03/2018
Yvan Devaux and 5 other authors -
Epigenetics in Ascending Thoracic Aortic Aneurysm and Dissection – 01/02/2018
Yvan Devaux and 3 other authors -
New dimensions in circadian clock function – 01/02/2018
Linda W. Van Laake and 2 other authors -
A heart-enriched antisense long non-coding RNA regulates the balance between cardiac and skeletal muscle triadin – 01/02/2018
Yvan Devaux and 4 other authors -
Atrial Structural Remodeling Gene Variants in Patients with Atrial Fibrillation – 01/01/2018
Philippe Chevalier and 9 other authors -
The circular RNA MICRA for risk stratification after myocardial infarction – 01/12/2017
Yvan Devaux and 3 other authors -
Long non-coding RNAs in the atherosclerotic plaque – 01/11/2017
Yvan Devaux and 7 other authors -
The Function and Therapeutic Potential of Long Non-coding RNAs in Cardiovascular Development and Disease – 15/09/2017
Yvan Devaux and 7 other authors -
Microrna 150-5p improves risk classification for mortality within 90 days after acute ischemic stroke – 01/09/2017
Mira Katan and 7 other authors -
Protein S100 as outcome predictor after out-of-hospital cardiac arrest and targeted temperature management at 33 °C and 36 °C – 20/06/2017
Pascal Stammet and 21 other authors -
Circular RNAs in heart failure – 01/06/2017
Yvan Devaux and 7 other authors -
Reprint of – 01/06/2017
Gunnar H. Heine and 9 other authors -
Biomarkers for heart failure – 23/03/2017
Yvan Devaux and 2 other authors -
MicroRNA profiling of human intermediate monocytes – 01/03/2017
Gunnar H. Heine and 9 other authors -
The Effects of Acute and Chronic Exercise on Skeletal Muscle Proteome – 01/02/2017
Bernardo Assis Petriz and 6 other authors -
Hypoxia inhibits lymphatic thoracic duct formation in zebrafish – 22/01/2017
Isabelle Ernens and 4 other authors -
Transcriptome of blood cells as a reservoir of cardiovascular biomarkers – 01/01/2017
Yvan Devaux -
Single versus serial measurements of neuron-specific enolase and prediction of poor neurological outcome in persistently unconscious patients after out-of-hospital cardiac arrest – A TTM-trial substudy – 01/01/2017
Sebastian Wiberg and 20 other authors -
Incremental value of circulating MiR-122-5P to predict outcome after out of hospital cardiac arrest – 01/01/2017
Yvan Devaux and 14 other authors -
A multicentre European registry to evaluate the Direct Flow Medical transcatheter aortic valve system for the treatment of patients with severe aortic stenosis – 01/12/2016
Christoph K. Naber and 26 other authors -
Which future for circulating microRNAs as biomarkers of acute myocardial infarction? – 01/11/2016
Yvan Devaux and 1 other author -
High-sensitivity troponin-T as a prognostic marker after out-of-hospital cardiac arrest – A targeted temperature management (TTM) trial substudy – 01/10/2016
Patrik Gilje and 13 other authors -
Usefulness of Serum B-Type Natriuretic Peptide Levels in Comatose Patients Resuscitated from Out-of-Hospital Cardiac Arrest to Predict Outcome – 01/10/2016
Martin Frydland and 20 other authors -
Myocardial Infarction-Associated Circular RNA Predicting Left Ventricular Dysfunction – 13/09/2016
Yvan Devaux and 9 other authors -
Association of circulating MicroRNA-124-3p levels with outcomes after out-of-hospital cardiac arrest – 01/06/2016
Yvan Devaux and 15 other authors -
Real-world multicentre experience with the Direct Flow medical® repositionable and retrievable transcatheter aortic valve implantation system for the treatment of high-risk patients with severe aortic stenosis – 01/02/2016
Christoph K. Naber and 9 other authors -
Adenosine A1 receptor activation attenuates cardiac hypertrophy and fibrosis in response to α1-adrenoceptor stimulation in vivo – 01/01/2016
Sarah Lena Puhl and 7 other authors -
Predictive value of interleukin-6 in post-cardiac arrest patients treated with targeted temperature management at 33°C or 36°C – 01/01/2016
John Bro-Jeppesen and 17 other authors -
Use of coronary ultrasound imaging to evaluate ventricular function in Adult Zebrafish – 01/01/2016
Isabelle Ernens and 3 other authors -
Circulating microRNAs and outcome in patients with acute heart failure – 01/11/2015
Marie France Seronde and 15 other authors -
Rat aortic ring model to assay angiogenesis ex vivo – 20/10/2015
Isabelle Ernens and 3 other authors -
Implication des ARNs non codants dans l’infarctus du myocarde et le remodelage ventriculaire post-infarctus – 02/10/2015
Jennifer Zangrando -
Whole transcriptome microarrays identify long non-coding RNAs associated with cardiac hypertrophy – 01/09/2015
Yvan Devaux and 9 other authors -
Erratum – 25/08/2015
Pascal Stammet and 18 other authors -
Long noncoding RNAs in cardiac development and ageing – 22/07/2015
Yvan Devaux and 8 other authors -
Exercise attenuates inflammation and limits scar thinning after myocardial infarction in mice – 15/07/2015
Sarah Lena Puhl and 6 other authors -
Neuron-specific enolase as a predictor of death or poor neurological outcome after out-of-hospital cardiac arrest and targeted temperature management at 33°C and 36°C – 19/05/2015
Pascal Stammet and 18 other authors -
Identification of candidate long noncoding RNAs associated with left ventricular hypertrophy – 01/04/2015
Yvan Devaux and 6 other authors -
Genetic variants are not associated with outcome in patients with coronary artery disease and left ventricular dysfunction – 24/02/2015
Arthur M. Feldman and 19 other authors -
MicroRNAs – 11/02/2015
Yvan Devaux and 6 other authors
Related News
Forthcoming events
-

Lecture series – Epidemiology & Prevention 2025-2026
Epidemiology & Prevention
18/06/2026 10:00
-

🇬🇧 Global trends in cancer survival: insights from the CONCORD programme and implications for cancer control
Epidemiology & Prevention
Speaker: Dr Michel P. Coleman
18/06/2026 11:00
-

Lecture series – Causal inference methods for real-world data 2025/2026
Lecture Series – Causal Inference Methods For Real-World Data
01/07/2026 11:00
-

Clinical Research Luxembourg Conference
Speaker: various
29/09/2026 08:00

Job vacancies
-
Master’s Student in Nutrition Sciences and Agri-Food Systems (TB)
Department of Precision Health – Nutrition & Health Research Group