cancer metabolism group
The Cancer Metabolism Group aims to understand the anabolic and catabolic demands of mammalian cells with respect to their specific microenvironment and the key determinants that can unbalance metabolic homeostasis, thereby leading to disease.
Activities
Cancer metabolism:
Malignant transformation of cells goes along with profound metabolic rewiring to cope with increasing biomass and energy demands. The goal of our research is to understand these metabolic changes pinpointing metabolic bottlenecks that are specific to cancer cells and which, ultimately, might become effective targets for clinical oncotherapy.
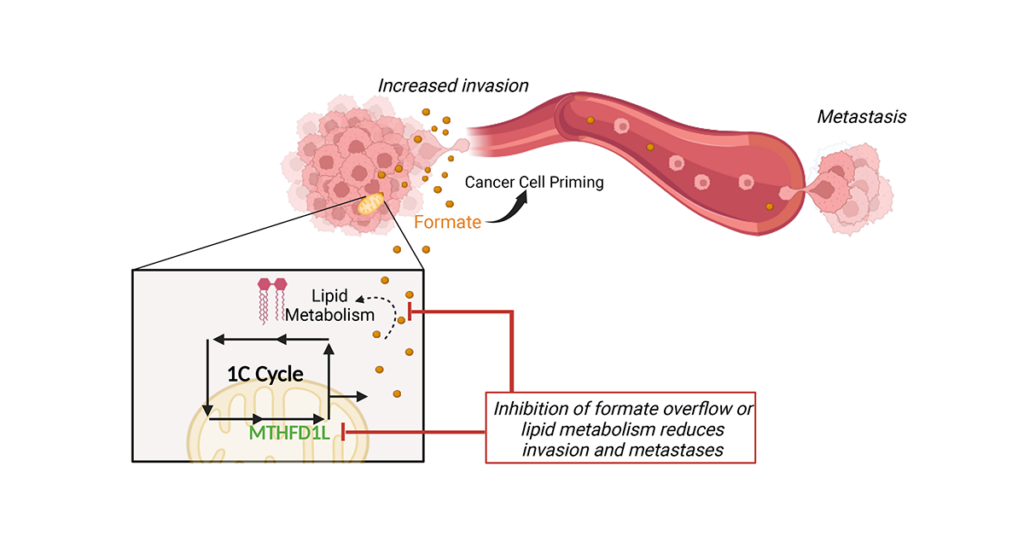
To measure the dynamics of cellular metabolism, we make use of stable-isotope tracers and high-resolution Mass Spectrometry. Applying these key tools with molecular and cell biology approaches allows a functional and mechanistic understanding of how metabolism impacts cancer cell proliferation, survival and dissemination. A particular focus resides at understanding the metabolic processes that allow cancer cell motility and invasion, a process essential for the onset of the metastatic cascade. Indeed, metastasis is accountable for 90 % of all cancer deaths. Yet, it remains one of the biggest challenges in cancer therapy and is currently seen as incurable.
An example of our work includes the study of a phenomenon known as “formate overflow”, a metabolic pathway which we have identified to influence cell motility and metastasis.
Tumour environment:
An additional area of interest is the interaction of cancer cells with their environment. Our work in this respect focuses on immune cells, as they play an important role in shaping either a tumour-promoting or -suppressive environment. Since metabolism plays an important role during immune cell activation, we aim to understand these metabolic dependencies and seek alternative immune cell-dependent entry points to target cancer cells.

Meiser
Partners
Cooperation Partners






Projects & clinical trials
Featured team members
Scientific publications
-
Development of a highly differentiated rat brain organoid model for exploring glioblastoma invasion dynamics and therapy – 24/11/2025
-
Formate – 04/10/2025
-
Fusobacterium nucleatum interacts with cancer-associated fibroblasts to promote colorectal cancer – 22/08/2025
-
Adaptation to Arginine Deprivation Leads to a More Aggressive, Therapy-Resistant Phenotype in HNSCC Cells – 19/06/2025
-
Modulation of the ACOD1/itaconate pathway differentially affects atherosclerosis severity across genetic models and sexes – 12/06/2025
-
NAD kinase essentiality in cancer – 02/05/2025
-
Calcium-mediated mitochondrial energy deficiency in Parkinson’s and Alzheimer’s diseases – 10/01/2025
-
Ketogenic diet suppresses colorectal cancer through the gut microbiome long chain fatty acid stearate – 20/02/2025
-
Protocol using ex vivo mouse brain slice culture mimicking in vivo conditions to study tumor growth and cell motility of glioblastoma cells – 18/10/2024
-
Presenting metabolomics analyses – 25/04/2024
Related News

Job vacancies
There are no jobs matching this page at the moment. You can view all jobs via the button below.