NORLUX scientist shines at EuroPDX conference
The EuroPDX Consortium is an initiative of translational and clinical researchers from 18 academic cancer centres and universities across Europe and in the United States, with the common goal of creating a network of clinically relevant models of human cancer, and in particular patient-derived xenograft (PDX) models. The long-awaited third edition was finally held in Egmong aan Zee in the Netherlands from the 27-29 of June 2022.
Anaïs Oudin, Research Engineer at the NORLUX Neuro-Oncology Laboratory, represented the team in an oral presentation entitled “Patient-derived orthotopic xenografts of Glioblastoma: how to recapitulate clinically relevant tumor microenvironment and treatment regimens” during the session on orthotopic PDX models. In particular, Anaïs focused on recapitulating clinically relevant tumour microenvironment and standard-of-care treatment protocols in animal models (see below for full abstract). Her work on patient-derived orthotopic xenograft models in glioblastoma was crowned when she was selected as the winner for best oral presentation for that session.
Congratulations Anaïs!
Patient-derived orthotopic xenografts of Glioblastoma: how to recapitulate clinically relevant tumor microenvironment and treatment regimens
Anaïs Oudin
Introduction
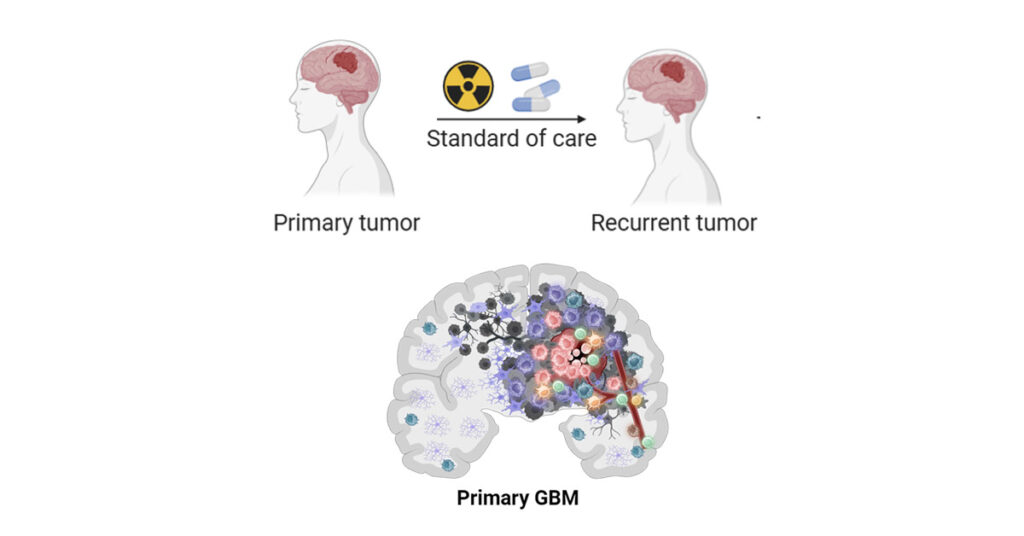
Glioblastoma (GBM) is the most deadly primary brain tumor despite a very aggressive treatment, which consists of surgical resection followed by radiotherapy and chemotherapy. Most GBM clinical trials fail due to inappropriate selection of the compounds at the preclinical stage. Therefore, appropriate preclinical models are crucial for achieving better treatment outcomes. GBM patient-derived othotopic xenografts (PDOXs) faithfully recapitulate the molecular and histopathological profiles of patient tumors. Orthotopic implantation allows for analogous tumor microenvironment and drug distribution though the blood brain barrier. However, current xenograft models suffer from the reduction of immune system components, which represents a bottleneck for adequate immunotherapy testing. Recapitulation of the standard-of-care regimen is challenging and it remains to be seen whether procedures combining surgical resections with clinically-relevant treatment protocols are practicable.
Material and Methods
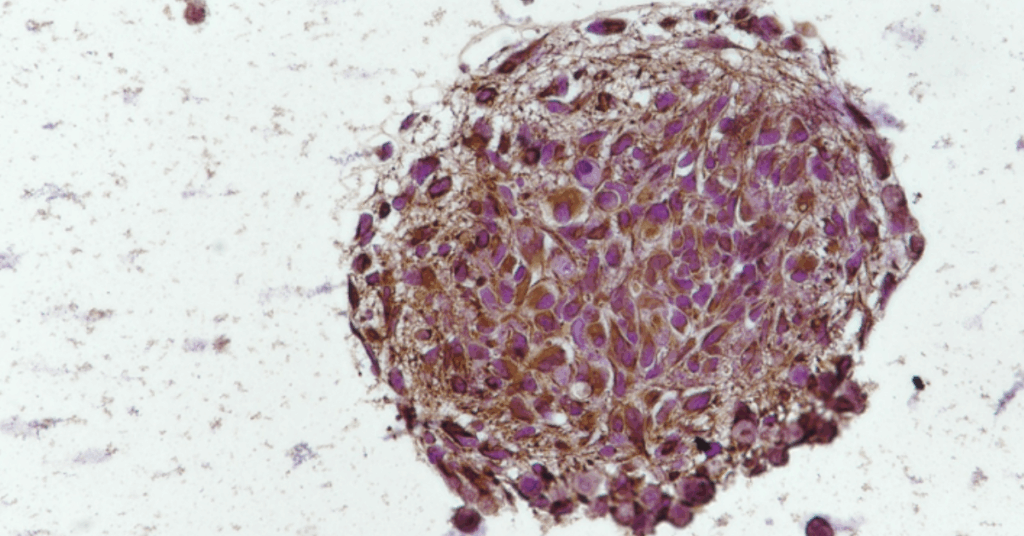
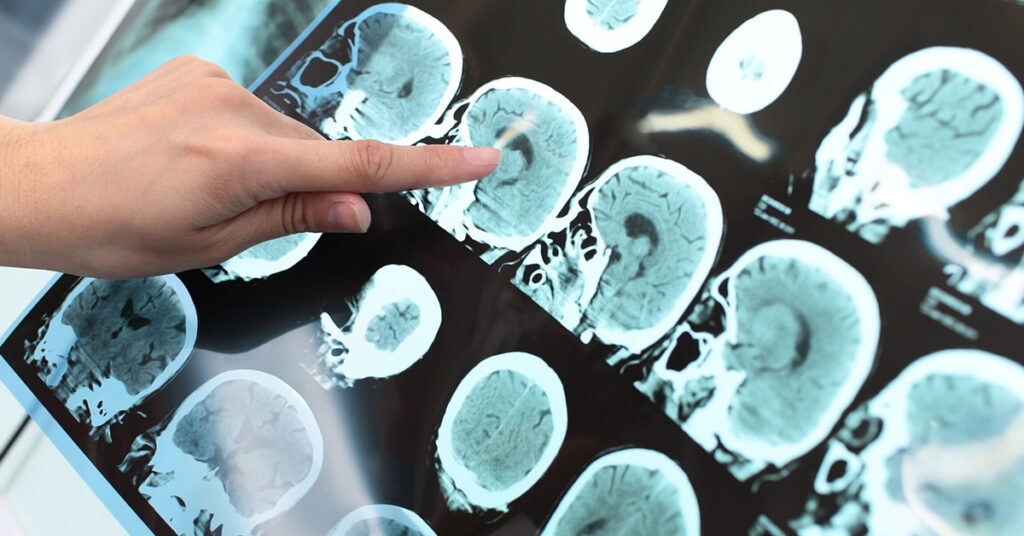
We regularly derive GBM PDOXs by intracortical implantation of primary patient-derived organoids in immunodeficient mice (NSG, Nude). To introduce the human adaptive immune system, GBM organoids were implanted in the brains of the human CD34+ hematopoietic stem cell-engrafted NSG (HU-CD34+) mice. Tumor growth was followed by Magnetic Resonance Imaging. Detailed characterization of the tumor microenvironment was performed by single cell transcriptomics, immunohistochemistry and multicolor flow cytometry. To mimic tumor recurrence, we performed surgery and tumor resection at the early tumor development stage. Robust in vivo treatment protocols were established for standard-of-care, novel drugs and combinatory treatments.
Results and Discussion
We show that PDOX models recapitulate major components of the tumor microenvironment found in human GBM, including microglia, astrocytes, oligodendrocytes and endothelial cells. Mouse cells reciprocally crosstalk with human GBM cells and switch towards GBM-instructed phenotypes. Missing adaptive immune system can be introduced by tumor growth in HU-CD34+ mice. HU-CD34+ mice present more than 50% of human immune cells in the bone marrow and blood. We observed an influx of human immune cells to the tumors developed in the brain. We further show that surgical resection can be applied on mouse brain and GBM tumors regrow after surgery similarly to patient GBM, leading to delayed survival time. Magnetic Resonance Imaging allows for robust assessment of the tumor growth and drug responses in vivo.
Conclusion
Our data provide insights into the recapitulation of the heterogeneous tumor microenvironment instructed by GBM cells in PDOXs. This work further confirms the wide applicability of GBM PDOXs for recapitulation of standard-of-care protocols and testing of novel treatments including immunotherapeutics.