News
LIH-made compound leads to discovery on how drug targets work
LIH383 helps to elucidate key mechanism in primary drug target
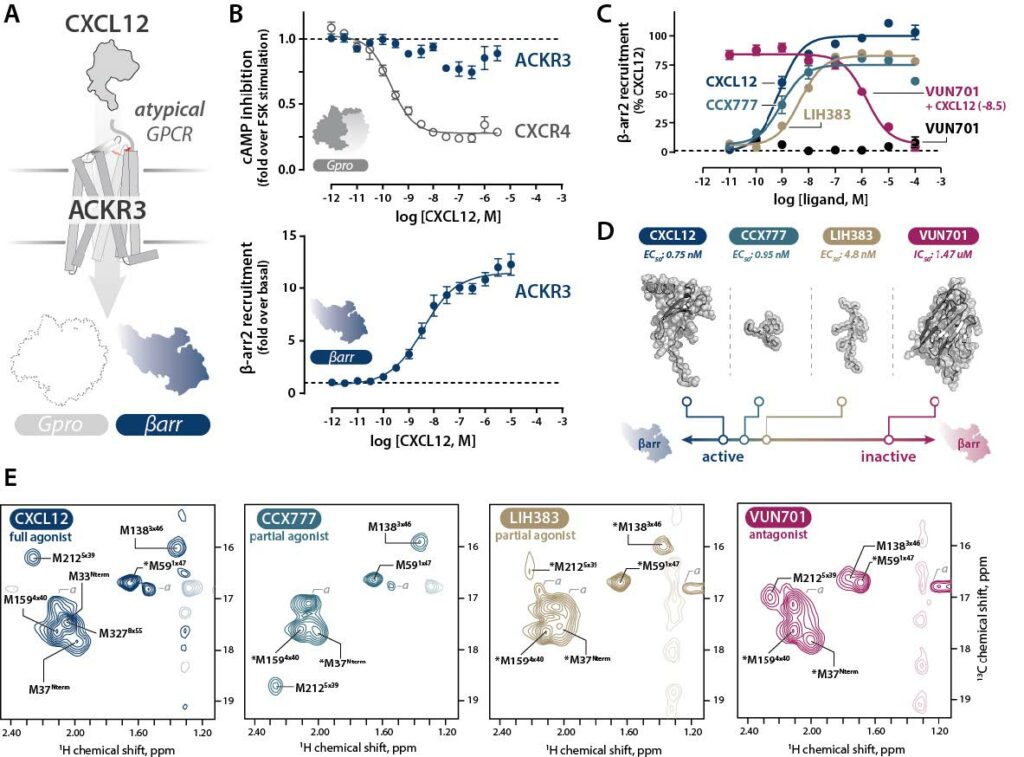
Luxembourg Institute of Health molecule LIH383, originally developed in the context of pain relief, was used to specifically target G protein-coupled receptors (GPCRs) to understand how these are regulated. GPCRs are targets of over a third of marked drugs. The study, led by the University of Wisconsin, elucidated previously unknown mechanisms for this class of receptors, thus opening new avenues to the development of more targeted therapies.
G protein-coupled receptors (GPCRs) lie on the surface of cells. Their main function is to detect and bind molecules that are circulating in their surroundings and, according to the molecule, transmit the designated ‘message’ to the inside of the cells. By binding to GPCRs, molecules can cause cells to initiate all sorts of processes. Amongst others, GPCRs are responsible for regulating our ability to see, taste and smell, as well as our blood pressure, heart rate, digestion and the proper functioning of our immune system.
β-arrestins are a type of protein that are found on the inside of cells. As their names suggests, they can bind to and ‘arrest’ or ‘block’ the activity of GPCRs, stopping or regulating whichever signal was activated from the outside. For a long time, it was thought that this was the only function of β-arrestins, but it was recently found that these could also control signalling pathways that are independent of G-proteins. New insights also revealed that some GPCRs are able to preferentially recruit β-arrestins to regulate one pathway selectively, and therefore a specific functional outcome, according to the molecule, or the ligand, binding to them. As more than 30% of US Food & Drug Administration (FDA)-approved drugs act through GPCRs, elucidating how they can actively recruit β-arrestins to regulate their cellular processes has important implications for drug development.
To shed light on this aspect, a group of researchers from the Medical College of Wisconsin used a type of GPCR that exclusively bind to β-arrestins, known as ‘atypical chemokine receptors’ (ACKRs), and, more specifically, ACKR3. The researchers then used Luxembourg Institute of Health (LIH) lead molecule LIH383 to target it, a compound that was originally developed for its selective ability to bind ACKR3 by LIH researchers Dr Andy Chevigné, Dr Martyna Szpakowska and Max Meyrath from the Department of Infection and Immunity.
Using nuclear magnetic resonance, reinforced by data generated with the LIH NanoLux platform to provide detailed structural analysis, the group found that ACKR3 can change conformation both on its ‘outside’ (the part of the receptor protruding from the cell) and ‘inside’ (the part of the receptor that mediates intracellular signalling) to selectively bind β-arrestins and therefore regulate cellular functional responses. The collaboration thus revealed previously unknown receptor dynamics, in addition to identifying the key regions responsible for the structural changes of GPCRs.
These findings provide not only a more detailed understanding of how GPCRs function, but also new insights on how they are regulated by small drugs,
says Dr. Martyna Szpakowska of the Immuno-Pharmacology and Interactomics group of the LIH.
GPCRs are crucial in health and disease, and, most importantly, are one of the major drug targets in medicine. Understanding exactly how they work allows us to target them more specifically, and therefore create medicines that are more efficient,
concludes Dr. Andy Chevigné.
The cutting-edge study, led by Dr. Andrew B. Kleist, Shawn Jenjak and Prof. Brian F. Volkman of the Medical College of Wisconsin, included a collaboration with the Immuno-Pharmacology and Interactomics group of the LIH, the UK MRC Laboratory of Molecular Biology, the Dutch Vrije Universiteit Amsterdam and US St. Jude Children’s Research Hospital. The findings were recently accepted in the esteemed journal Science under the full title: “Conformational selection guides β-arrestin recruitment at a biased G protein-coupled receptor.”
About LIH383
LIH383 is a LIH proprietary molecule that was developed and patented by the team of Immuno-Pharmacology and Interactomics group in the Department of Infection and Immunity at the Luxembourg Institute of Health, during their work with opioid regulation in 2020 (Meyrath et al. Nature Communications, 2020). The lead compound is able to bind specifically to an atypical opioid receptor called ACKR3, and consequently prevent it from capturing naturally secreted opioids and increase their availability in the brain. It is expected that LIH383 will lead to the development of a new class of drugs for pain and depression, providing a novel and innovative therapeutic strategy to address the opioid crisis. More information can be found here.